Immunotherapy for Cancer
Doctors and scientists at the OHSU Knight Cancer Institute are national leaders in harnessing the immune system’s natural ability to fight cancer. We have worked for a decade to help develop new therapies, first for blood cancers and now rapidly expanding to other cancer types.
We have many clinical trials to test promising new immunotherapies, and more on the way. Our goal is to make these next-generation therapies available to patients across the Northwest.
- OHSU was the first hospital in the Northwest to offer the CAR-T cell therapy Kymriah.
- In March 2021, we became the first center in the world to use NK therapy, a type of immunotherapy, in a patient with advanced lymphoma.
- Our doctors are studying whether immune therapy medications called checkpoint inhibitors can help men with advanced prostate cancer.
- The cancer research network based at OHSU is leading the first federally funded clinical trial of immunotherapy for rare cancers.
- The Knight Cancer Institute was among the first in the nation to treat patients who have advanced prostate cancer with the immunotherapy medication Provenge.
Learn more about our nationally recognized doctors, on our Bone Marrow and Stem Cell Transplant page.

What is immunotherapy?
As tumors develop, they find ways to hide from the immune system and avoid its defenses. Immunotherapies change how the immune system works so it can find and destroy cancer cells.
This approach can have longer-lasting effects than traditional chemotherapy. Immunotherapies have greatly increased survival times in people with aggressive cancers such as advanced melanoma and advanced lung cancer.
There are many types of immunotherapy. Treatments can be used alone or combined with other cancer therapies. Immunotherapies may be given in an IV, shot, pill or other form.
Types of immunotherapy at OHSU
Here are examples of cancer-fighting immunotherapies available at the Knight Cancer Institute. Some of these therapies have been approved by the Food and Drug Administration, and others are available to eligible patients through clinical trials.
CAR T-cell therapy
T cells are immune cells that circulate in the body to fight infection. In CAR T-cell therapy, some of the patient’s T cells are collected from the patient’s blood.
A lab then genetically modifies the T cells so they develop a special type of protein that binds to and kill cancer cells. These proteins are called chimeric antigen receptors (CARs).
The lab grows millions of these modified T cells and sends them back to the hospital. The cells are returned to the patient’s bloodstream in an IV drip to find and destroy cancer.
Learn more about CAR T-cell therapy and our team’s role in developing the first CAR T therapy approved by the FDA.
NK cell therapy
A patient’s NK (natural killer) cells — like T cells — can be modified to develop chimeric antigen receptors (CARs) that target cancer cells. NK cells can also be collected from donor blood or umbilical cord blood and given, unchanged, to patients.
Scientists are investigating other properties of NK cells to fight cancer. Compared with T cells, NK cells have some advantages and more versatility.
Our team became the first in the world to use an NK therapy from Artiva Biotherapeutics in a patient with advanced lymphoma. We designed and are leading the clinical trial for this therapy. It’s one of the NK therapies in clinical trials for leukemia and lymphoma.
Checkpoint inhibitors
Tumors can slow or stop immune defenses by sending false signals to the immune system’s checkpoints. Medications called checkpoint inhibitors release the brakes to unleash full-force immune defenses.
A checkpoint inhibitor called ipilimumab was the first treatment to improve survival in advanced melanoma. Now many checkpoint inhibitors have proved helpful in treating several types of cancer.
All are given by IV and require an experienced care team, such as the one at the Knight Cancer Institute, to prevent and handle side effects that can be severe.
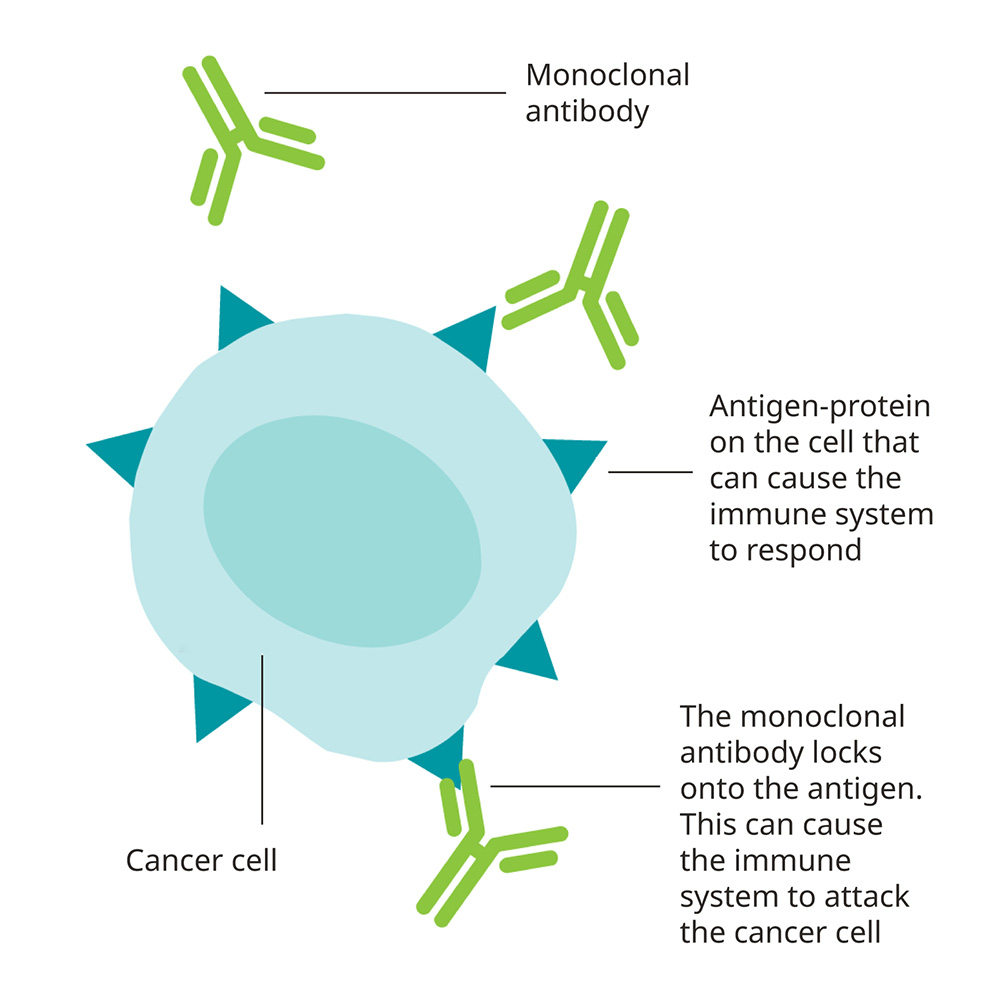
Monoclonal antibodies
Monoclonal antibodies are lab-made proteins that work like the natural antibodies of the immune system. They stick to specific targets, such as protein on the outside of a cancer cell.
Some monoclonal antibody treatments are designed to block signals that cancer cells use to grow. This is a kind of targeted therapy. Other monoclonal antibody therapies are used as checkpoint inhibitors, boosting immune defenses by blocking immune system checkpoints.
Monoclonal Antibody Therapy
BiTE therapy
This is a type of monoclonal antibody therapy. It uses a monoclonal antibody called a BiTE (bispecific T-cell engager). The BiTE forms a link between a cancer cell and a T cell, helping the T cell recognize the cancer cell and kill it.
A BiTE medication called Blincyto (blinatumomab) is FDA-approved for use against types of acute lymphoblastic leukemia (ALL).
Cancer vaccines
In the same way that ordinary vaccines prime the immune system to stop infections, cancer vaccines prompt the immune system to attack cancer cells.
Some cancer vaccines treat cancer or stop it from coming back. Others prevent the types of cancers that can be triggered by viral infections.
For example:
- Provenge (sipuleucel-T) is used to treat advanced prostate cancer.
- Gardasil 9, an HPV (human papillomavirus) vaccine, can prevent infections that are linked to cervical cancer, penile cancer, cancers of the mouth and throat, and other types.
Cytokines
Cytokines are proteins that serve as messengers between cells. They also help control immune system responses. Used as a treatment, cytokines boost the growth of immune cells that can attack cancer.
- Interleukin-2, or IL-2, is used to treat advanced melanoma and kidney cancer.
- Interferon-alfa is used to treat kidney cancer, melanoma, and some kinds of leukemia and lymphoma.
TIL therapy
TIL stands for tumor-infiltrating lymphocytes. Lymphocytes are white blood cells that fight infection as part of the immune system. They attack cancer, too, but sometimes the cancer fights back and shuts them down.
In this therapy, doctors remove some of your lymphocytes, turn them back on, make lots of copies, and give them back to you. Find out more about TIL therapy at OHSU.
IVIG therapy
Intravenous immune globulin is made of infection-fighting antibodies collected from thousands of blood donors. It’s given by IV drip. Patients with multiple myeloma or chronic lymphocytic leukemia may receive it to help their bodies ward off infections.
Immunotherapies by cancer type
The FDA has approved immunotherapy treatments for many kinds of cancers, including some of the most common cancers.
Not all patients with these cancers are eligible for immunotherapy. It depends on factors such as how far the cancer has advanced, what kind of mutations it has, and how it has responded to other treatments.
Breast cancer
Some breast cancers can be treated with an immune checkpoint inhibitor called Tecentriq (atezolizumab), a monoclonal antibody given by IV drip. Breast cancers that make too much of a protein called HER2 can be treated with monoclonal antibodies that target and block HER2 signaling.
Colon and rectal cancer
Checkpoint inhibitors may be used in patients with colon and rectal cancers that show specific gene changes. They include these, all given by IV:
- Keytruda (pembrolizumab)
- Opdivo (nivolumab)
- Yervoy (ipilimumab)
Leukemia and lymphoma
CAR T-cell therapies have transformed the treatment of some forms of leukemia and lymphoma. The patient’s immune T cells are removed, modified to target cancer, and put back in the bloodstream. Some blood cancers are also treated with monoclonal antibodies.
The FDA has approved CAR T-cell therapies to treat:
- Some forms of acute lymphoblastic leukemia (ALL)
- Follicular lymphoma (FL)
- Large B-cell lymphoma
- Mantle cell lymphoma
Melanoma
Immune checkpoint inhibitors were the first treatment to improve survival in people with melanoma that has spread to other parts of the body. The FDA has approved several, including:
- Keytruda
- Opdivo
- Tecentriq
- Yervoy
All are given by IV drip to treat melanomas that can’t be removed by surgery or that have spread to other parts of the body.
Cytokine therapies also are used to treat some patients with melanoma.
We also offer tumor-infiltrating lymphocyte therapy (TIL therapy) for people with late-stage melanoma.
Multiple myeloma
The FDA has approved a CAR T-cell therapy, Abecma (idecabtagene vicleucel), to treat adults with advanced multiple myeloma. Three monoclonal antibody therapies are also approved for types of this cancer.
In clinical trials, scientists are making strides in using NK therapy, BiTE therapy and other immunotherapies for multiple myeloma.
Prostate cancer
Provenge is a cancer vaccine made from a patient’s own immune cells to trigger defenses against prostate cancer. The checkpoint inhibitor Keytruda is FDA-approved for some patients with advanced prostate cancers that show specific gene changes.

Clinical trials and research
Cancer patients at OHSU have access to the latest immunotherapy treatment advances. They include experimental treatments unavailable elsewhere in the region.
Knight Cancer Institute doctors are leading the development of new immunotherapies and finding ways to use immunotherapies against more cancer types.
Clinical trials: We offer clinical trials that give eligible patients a chance to try experimental treatments. Your care team will talk with you about whether a trial is right for you.

Research:
- CAR T-cell therapy for lymphoma not only extends survival, it may improve quality of life after treatment, according to research led by OHSU's Richard Maziarz, M.D. Dr. Maziarz is medical director of the Knight Cancer Institute’s adult blood and marrow stem cell transplant and cellular therapy program.
- Dr. Jennifer Saultz is leading the clinical trial in which the first person in the world received an NK therapy from Artiva Biotherapeutics for advanced lymphoma. The trial is investigating the effectiveness of this therapy against a range of non-Hodgkin lymphomas.
- Discoveries by our scientists are paving the way to using immune checkpoint inhibitors to treat acute myeloid leukemia. AML is the most common acute blood cancer in adults, and one of the most difficult to treat.
- Our researchers have uncovered how tumor cells trick the immune system into creating inviting conditions for new, medication-resistant tumors to grow.
- Knight Cancer researchers are using gut bacteria and the immune system to fight tumors. They are testing the idea that changing the mix of bacteria living in the intestines could make aggressive prostate cancers more responsive to treatment with immune therapy medications called PD-1 inhibitors.
- The SWOG Cancer Research Network, based at OHSU, is coordinating a clinical trial funded by the National Cancer Institute to test an immunotherapy approach for more than 30 rare cancers.
- OHSU was the first in the Northwest to offer Kymriah, the first CAR T-cell therapy approved by the FDA, for one of the most common and lethal forms of cancer in children and young adults.
- OHSU researchers are doing studies on CAR T-cell therapy to see if T cells from donors — instead of from the patient — are more effective at killing cancer cells.
- Results from clinical trials at OHSU and other sites showed high rates of long-lasting responses to CAR T-cell therapy for an aggressive lymphoma.
- Our scientists work in the innovative Knight Cancer Research Building, a 320,000-square-foot center designed to encourage collaboration.
Learn more
- Immunotherapy to Treat Cancer, National Cancer Institute
- Immunotherapy, American Cancer Society
- How Immunotherapy Is Used to Treat Cancer, American Cancer Society
- Understanding Cancer Clinical Trials, OHSU Knight Cancer Institute
For patients
Call 503-494-7999 to:
- Request an appointment
- Seek a second opinion
- Ask questions
Help and education:
Location
Knight Cancer Institute, South Waterfront
Center for Health & Healing, Building 2
3485 S. Bond Ave.
Portland, OR 97239
Free parking for patients and visitors
Refer a patient
- Refer your patient to OHSU.
- Call 503-494-4567 to seek provider-to-provider advice.
Kids and teens

Read more
Learn more about OHSU Knight Cancer Institute treatments: