CAR T-Cell Therapy for Cancer
T cells are immune system warriors that circulate in blood to fight infections. CAR T-cell therapy is an inventive treatment that takes T cells from the patient’s body and modifies them to destroy cancer cells.
CAR T-cell therapy, a type of immunotherapy, is a particular breakthrough for children with leukemia, and for adults with certain hard-to-treat blood cancers. It also holds promise for other diseases.
Doctors at the OHSU Knight Cancer Institute have been at the forefront of clinical trials of this therapy. We’re building an expert team to make this complex treatment available to patients across the region.
- OHSU was the first hospital in the Northwest to offer patients Kymriah, the first CAR T-cell therapy approved by the Food and Drug Administration.
- A Knight Cancer Institute doctor co-led the first study to outline Kymriah’s long-term effectiveness.
- Our doctors helped lead the clinical trial showing that CAR T-cell therapy for lymphoma not only extends survival but may improve quality of life after treatment.
Read more about:
- CAR T-cell therapy for children
- Our team’s exceptional expertise, on our Bone Marrow and Stem Cell Transplant page
- Immunotherapy at the Knight Cancer Institute
Understanding CAR T-cell therapy
CAR T-cell therapy is a complex, multistep treatment. It’s best handled at centers — such as the Knight Cancer Institute — with deep experience doing stem cell and bone marrow transplants. Our team has done more than 4,500 transplants, putting us among the nation’s top 20 transplant centers.
What is CAR T-cell therapy?
Let’s start with some definitions.
- T cells are lymphocytes — a type of white blood cell that fights infection as part of the immune system.
- CAR (chimeric antigen receptor) is a new kind of protein. “Receptor” means it can bind to a cancer cell, like a key fitting in a lock.
- CAR T-cell therapy is a gene therapy because genes in the patient’s T cells are reprogrammed to make CARs.
CAR T-Cell Therapy
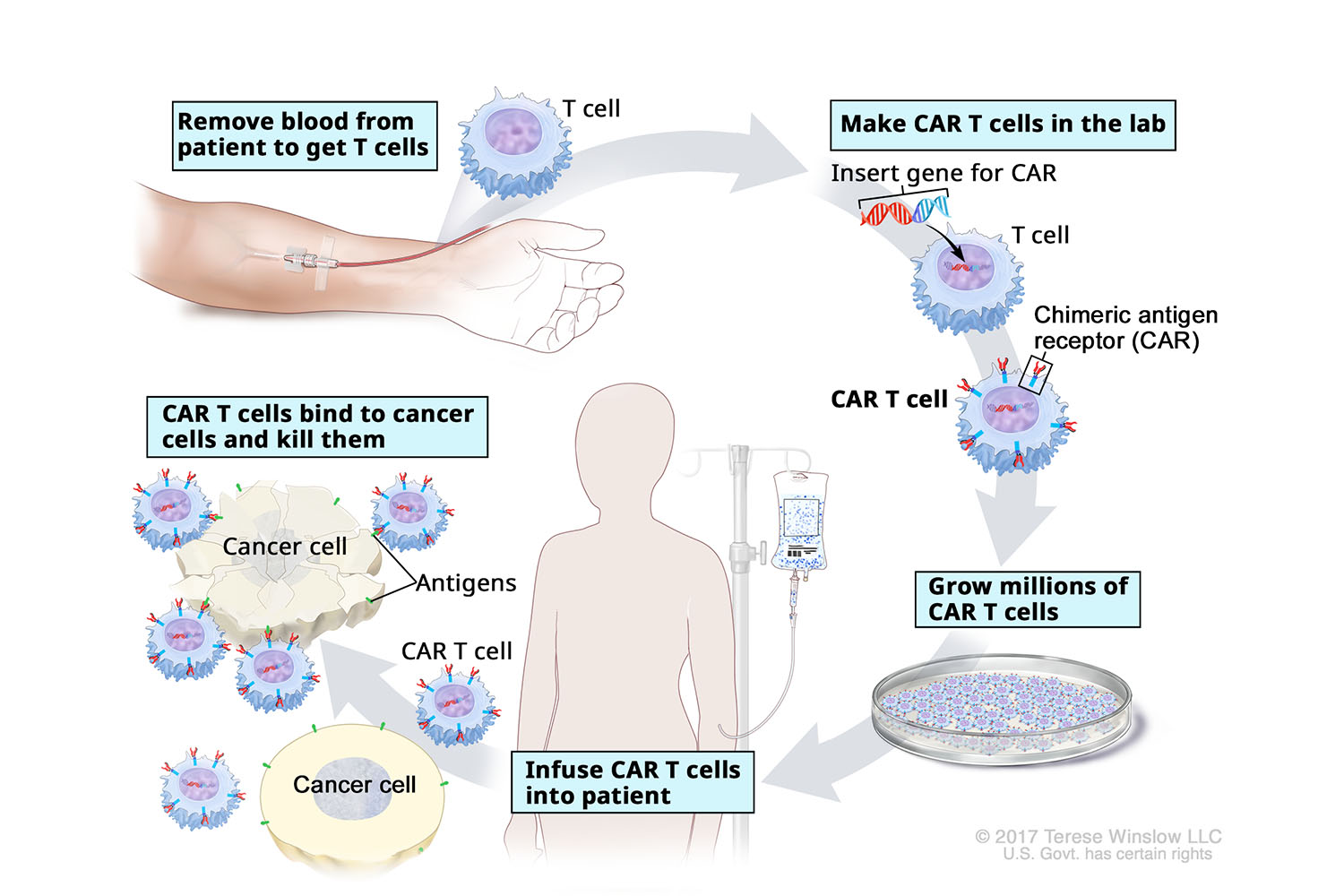
How CAR T-cell therapy works
How T cells are collected: Treatment starts with collecting T cells from the patient’s blood. Blood is drawn through one IV line and filtered to collect T cells. The rest is returned through another IV line.
How T cells are modified: The T cells are sent to a lab where they are genetically modified to make the chimeric antigen receptor, or CAR. This protein binds to specific proteins on the surface of cancer cells. That equips the modified T cells to target and kill the cancer cells.
Treatment: The lab grows millions of the modified T cells over several weeks, and ships them back to the hospital. Doctors may give the patient a short course of chemotherapy before giving them the CAR T cells in an IV drip. Once in the patient’s bloodstream, the CAR T cells multiply and may be able to destroy all the cancer cells.
Results of CAR T-cell therapy
The therapy has led to long-term remission for children and adults with some types of leukemia and lymphoma. The reprogrammed cells may stay in the body to fight cancer for years.
Side effects
Some people have had serious side effects, including high fevers and dangerously low blood pressure. Other serious side effects include brain swelling, confusion, seizures and severe headaches. Experienced centers such as the Knight Cancer Institute have learned how to recognize and treat side effects early.
Research
OHSU researchers are studying using donor T cells — instead of the patient’s own cells — for CAR T-cell therapy. Donor cells may be more effective at fighting cancer. We are also researching how to use this therapy for other diseases, such as sarcoma.
Cancers with FDA-approved CAR T-cell therapies
The Food and Drug Administration (FDA) has approved six CAR T-cell therapies:
- Abecma (idecabtagene vicleucel)
- Breyanzi (lisocabtagene maraleucel)
- Kymriah (tisagenlecleucel)
- Tecartus (brexucabtagene autoleucel)
- Yescarta (axicabtagene ciloleucel)
- Carvykti (ciltacabtagene autoleucel)
By cancer type, they are approved to treat:
Acute lymphoblastic leukemia (ALL): Kymriah is approved for patients up to age 25 with B-cell precursor acute lymphoblastic leukemia that resisted treatment or came back after treatment (relapsed). ALL is one of the most common and lethal forms of cancer in children and young adults.
B-cell lymphoma: Yescarta, Kymriah and Breyanzi are approved for adults with large B-cell lymphoma that resisted two or more therapies or that relapsed. This aggressive blood cancer is the most common type of non-Hodgkin lymphoma.
Follicular lymphoma (FL): Yescarta is approved to treat adults (18 and older) with follicular lymphoma that has resisted treatment, or came back, after two other therapies.
Mantle cell lymphoma: Tecartus is approved for adults with treatment-resistant or relapsed mantle cell lymphoma.
Multiple myeloma: Abecma and Carvykti are approved to treat adults (18 and older) with multiple myeloma that did not respond, or that returned, after four other therapies.
CAR T-cell therapy for children
Cell therapies can boost your child’s own immune system to help it recognize and kill cancer cells. The pediatric cancer team at OHSU Doernbecher Children’s Hospital is a leader in offering these treatments.
The team includes doctors, advanced practice providers, nurses, dietitians, social workers and other specialists working together to care for your child and family.
We’ve done thousands of cell therapy procedures, including CAR T-cell therapy for children with leukemia.
Learn more
- CAR T Cell Therapy, Be the Match
- CAR T cells: Engineering Patients’ Immune Cells to Treat Their Cancers, National Cancer Institute
- Chimeric Antigen Receptor (CAR) T-Cell Therapy, Leukemia & Lymphoma Society
- CAR T-cell Therapy and its Side Effects, American Cancer Society
- Immunotherapy for Cancer at the Knight Cancer Institute
- Leukemia treatment at the Knight Cancer Institute
- Lymphoma treatment at the Knight Cancer Institute
For patients
Call 503-494-7999 to:
- Request an appointment
- Seek a second opinion
- Ask questions
Help and education:
Location
Knight Cancer Institute, South Waterfront
Center for Health & Healing, Building 2
3485 S. Bond Ave.
Portland, OR 97239
Free parking for patients and visitors
Refer a patient
- Refer your patient to OHSU.
- Call 503-494-4567 to seek provider-to-provider advice.
Kids and teens
