Lampi Lab
Our focus is on investigating the role of deamidation in protein dynamics using multiangle laser scattering and mass spectrometry.
Our research
The Lampi Laboratory has spent over 25 years investigating the role of deamidation in protein dynamics using multiangle laser scattering and mass spectrometry. Deamidation, which introduces a negative charge in a protein, alters the biophysical properties of the major proteins in the eye lens, crystallins. We have applied static and dynamic light scattering to determine soluble aggregate sizes and hydrogen-deuterium exchange with high-resolution mass spectrometry to identify changes in protein solution dynamics. Using these methods, β-crystallins were determined to be flexible and take on different conformations depending on the partner subunit, representing a fundamental change in how we view these structural proteins. We have also shown that deamidation disrupts interactions between β-crystallin subunits, increases the propensity of crystallins to form large molecular weight aggregates, and prevents rescue by the a-crystallin chaperone. These results have led to our hypothesis that an accumulation of deamidated crystallins in the aged and cataractous lens is detrimental to lens function.
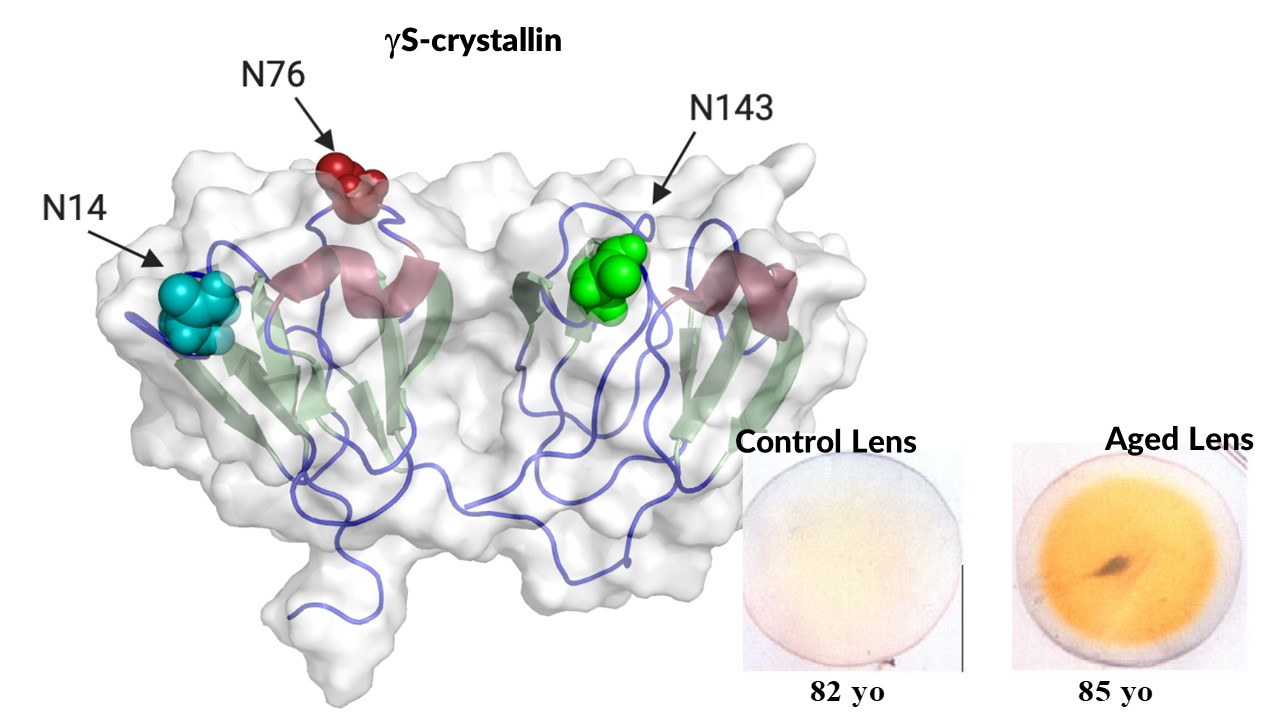
Together with our longtime colleague, Dr. Larry David, we have identified cumulative deamidation sites on the surface of gS-crystallin that increase the propensity for disulfide bonding and lock the protein into alternant aggregation prone structures. Furthermore, racemization and isomerization, two modifications that accompany the extensive deamidation of lens crystallins in age-related cataract, may potentially be more disruptive to protein structure than deamidation alone. However, the functional significance of these modifications in lens crystallins has never been determined. This new project will for the first time introduce these modifications at specific physiologically relevant sites in gS-crystallin so that the resulting consequence on protein structure can be determined.
Our lab members
Kirsten Lampi | Sam Wheeler
Funding
NIH R01 EY012239
NIH R01 EY027012
NIH R01 EY027768
NIH P30 EY10572
Longtime collaborators
Larry David
Oregon Health & Science University
Jim Prell
University of Oregon
Steve Reichow
Oregon Health & Science University
Eugene Shaknovich
Harvard
Om Srivastava
University of Alabama
Takumi Takata
Kyoto University
