Harding Metabolic Disorders Lab

The Harding Lab develops novel gene therapies to treat inherited errors of metabolism (IEMs).
For over 35 years, Dr. Harding and his team have studied the etiology, management, and treatment of metabolic disorders, such as phenylketonuria (PKU). These disorders are individually rare though as a group they occur in 1 in 2500 births. Treating these disorders often requires lifelong dietary restrictions and low-protein medical foods. In life threatening metabolic disorders, patients may need a liver transplant.

For example, patients with PKU can’t metabolize the amino acid phenylalanine due to mutations in the phenylalanine hydroxylase (Pah) gene. Doctors treat them with medical foods that contain all other amino acids while restricting protein in their diet. Without this treatment, patients, especially children, are at risk of permanent intellectual impairment.
But much remains unknown about the consequences of lifelong dietary restriction. At the Harding Lab, we hope to improve quality of life for these often-overlooked patients. We are exploring new ways to evaluate and treat patients and studying potential cures using gene editing.
You can:
Research and Publications
The Harding Lab studies the basic science of gene editing using cell culture and mouse models of PKU. We broadly define these gene editing approaches as:
- Gene insertion
- Gene addition
- Gene correction
The Harding Lab has published studies on all three technologies, but we focus on gene insertion due to its potential to create a one-size-fits-all treatment for each disorder. You can find more publications by Dr. Harding on PubMed.
Gene insertion
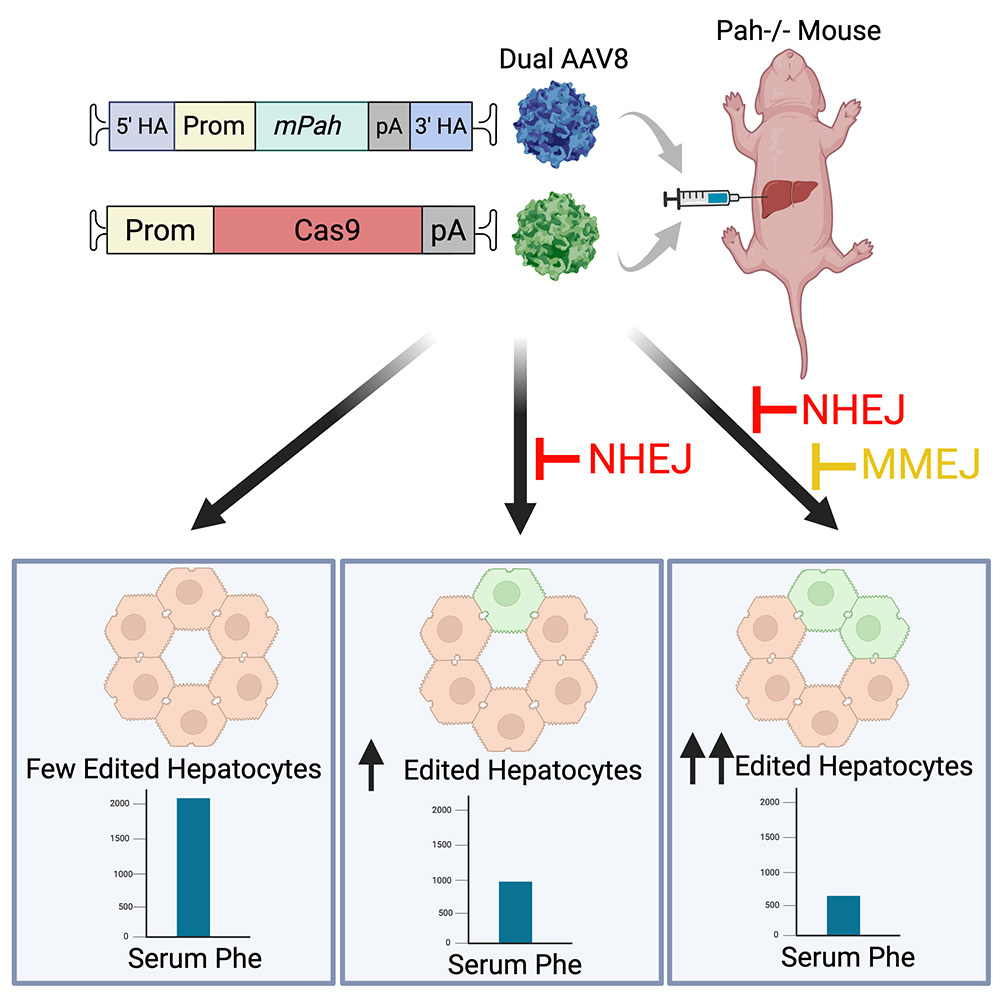
Gene insertion attempts to insert a complete and functional sequence of a gene into the genome of target cells. Unlike gene correction, this can permanently treat a monogenetic disease regardless of the specific mutation causing it. Unlike gene addition, treatment using this approach will last a lifetime. Our previous studies have reduced blood phenylalanine levels near the therapeutic threshold in mice with PKU.
We are studying ways to deliver the phenylalanine hydroxylase (PAH) gene into liver cells at a high enough rate to lower phenylalanine levels in the blood. One method uses a gene-editing tool called CRISPR/Cas9 Prime Editor to help insert the gene using a process called dual flap integration (DFI). Another method tests a new gene-editing strategy called Programmable Addition via Site-specific Targeting Elements (PASTE).
In both approaches, we use Prime Editor first to write a short targeting sequence at a specific place in the genome. In the DFI method, a therapeutic gene carrying the PAH DNA recognizes this sequence and inserts itself into the genome through recombination signals on each side of the gene. In the PASTE method, a protein called the BxbI integrase recognizes the target sequence created by Prime Editor and inserts a large piece of DNA at that site.
We are testing each step of this process using adeno-associated virus (AAV) and lipid nanoparticle delivery systems to bring the necessary gene-editing components into PAH-deficient mice.
Publication:
Enhancement of Therapeutic Transgene Insertion for Murine Phenylketonuria
Molecular Therapy 2026
Martinez M, Richards DY, Winn SR, Baris AM, Vonada A, Harper L, Harding CO
Gene addition
Gene addition attempts to provide a DNA expression cassette into the nucleus of target cells, but not into the patient’s own DNA. Our previous research in gene addition used the AAV8 viral vector to deliver a PAH expression cassette into mice with PKU. We measured their blood phenylalanine concentrations and cognitive ability over the course of their lives to inform future clinical studies. A drawback of this approach is that the treatment wears off over time due to the loss of viral episomes in dividing hepatocytes. Human clinical trials confirmed this drawback.
Publications:
Blood phenylalanine reduction corrects CNS dopamine and serotonin deficiencies and partially improves behavioral performance in adult phenylketonuric mice
Molecular Genetics and Metabolism 2018
Winn SR, Scherer T, Thöny B, Ying M, Martinez A, Weber S, Raber J, Harding CO
Complete correction of hyperphenylalaninemia following liver-directed, recombinant AAV2/8-vector-mediated gene therapy in murine phenylketonuria
Gene Therapy 2006
Harding CO, Gillingham MB, Hamman K, Clark H, Goebel-Daghighi E, Bird A, Koeberl DD
Gene correction
Gene correction attempts to correct a mutation to rescue functionality of the patient's DNA sequence. One method called homology-directed repair uses CRISPR/Cas9 to create a targeted double-strand break at the mutation site while providing a repair template.
An increasingly popular method of gene correction uses CRISPR base editors to convert one base pair into another without inducing double-strand breaks. A drawback of this approach is that each unique mutation needs unique reagents. This is problematic in disorders like PKU that can be caused by thousands of unique mutations.
Publication:
AAV-Mediated CRISPR/Cas9 Gene Editing in Murine Phenylketonuria
Molecular Therapy 2019
Richards DY, Winn SR, Dudley S, Nygaard S, Mighell TL, Grompe M, Harding CO
Meet our team
-
-
Appointments and titles
- Professor of Molecular and Medical Genetics, School of Medicine
-
Areas of interest
- Gene therapy
- Inborn errors of metabolism
- Metabolic disorders
- Phenylketonuria (PKU)
-
Shelley R. Winn, Ph.D., senior research associate

Since earning a Ph.D. from Brown University in the 1980s, Dr. Winn has gained vast experience in designing and cloning plasmids for use in viral and nonviral vector systems. Dr. Winn has collaborated on projects in musculoskeletal tissue regeneration, gene therapy and general tissue engineering, including early work to optimize processes for the isolation and procurement of progenitor/stem cells. Dr. Winn’s expertise and experience are critical to the Harding Lab's effort to develop a safe and permanent treatment for phenylketonuria (PKU) using liver-directed gene therapy.
Michael Martinez, Ph.D., postdoctoral researcher

Dr. Martinez earned a B.S. in biochemistry from Humboldt State University in 2017. After completing his undergraduate studies, Michael joined the laboratory of Dr. Anders Persson at UCSF as a California Institute of Regenerative Medicine (CIRM) Bridges Fellow. He studied the role of the IDH1 mutation in the formation of low-grade gliomas. In 2025, he earned a Ph.D. in Molecular and Medical Genetics at OHSU for his research investigating new strategies to improve gene editing. He continues his research in the Harding lab. In his free time, Michael enjoys playing tennis (poorly), grilling, and exploring the greater Portland area.
Laura J. Young, B.S., graduate student

Young earned her B.S. in genetics and genomics with a minor in quantitative biology and bioinformatics from UC Davis in 2022. As an undergraduate, Young worked on genetic research at the UC Davis School of Veterinary Medicine’s Comparative Ophthalmology and Vision Sciences Laboratory with Dr. Sara Thomasy. After graduation, she joined the research laboratory of Dr. Maija Kiuru at the UC Davis School of Medicine. There she led research to characterize and advance diagnosis of rare genetic skin conditions. Young is pursuing her Ph.D. in biomedical sciences at OHSU. Her research will focus on investigating novel gene editing modalities with the goal of developing a permanent lifelong treatment for PKU. In her free time, Laura enjoys exploring Portland’s coffee shops and making new vegetarian recipes.
Support our work
Help us find promising new treatments for PKU and other metabolic disorders.
Presentations
Hear about our work at:
Clinical studies
We are part of the PHEFREE Consortium, a clinical research group funded by NIH.
We are recruiting adults and children for a longitudinal study of hyperphenylalaninemia. This study aims to learn more about people who have conditions that raise phenylalanine levels, such as PKU. If you join the study, we will gather information about your health, well-being and thinking abilities over time.