Training and Education
Whether you’re a faculty member, fellow, student, study coordinator, mentor, or mentee, OCTRI offers a wealth of educational and training opportunities to hone your expertise in clinical and translational research.
-
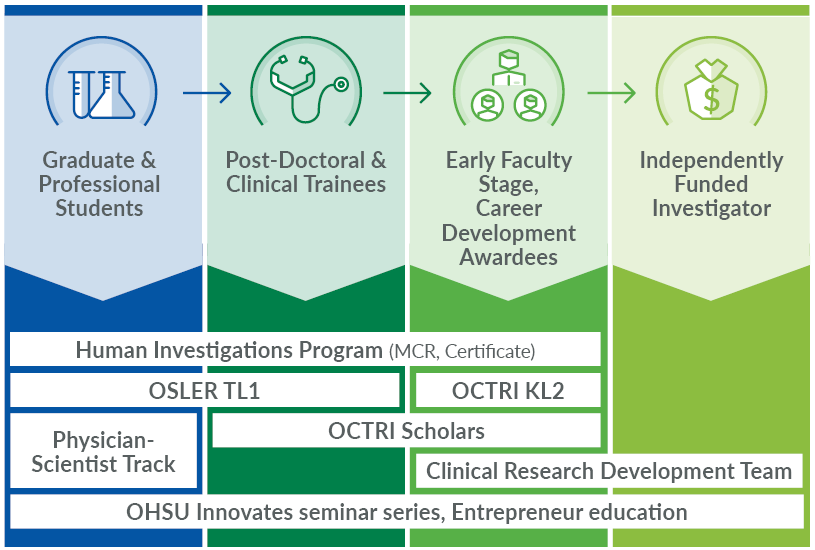
Are you an MD, DMD, PhD, PharmD, or DNP student who wants to expand your experience with clinical and translational research? Check out these opportunities from OCTRI.
Oregon Students Learn and Explore Research (OSLER) TL1 Program
Research experience and mentorship through stipend-funded training. Learn more on the TL1 Program web page.
Human Investigations Program (HIP)
An integrated, academic curriculum in clinical and translational education, featuring a Master of Clinical Research degree, Certificate in Human Investigations, and individual courses. Review options on the HIP web page.
Physician-Scientist Experience
The Physician-Scientist Experience provides OHSU medical students with the opportunity to explore translational medicine as a career choice in a 5-month or year-out mentored research experience. Visit the Physician-Scientist Experience web page.
OCTRI Research Forum
Seminar series highlighting practical topics of interest to clinical and translational researchers. Find upcoming seminars on the OCTRI Research Forum web page.
-
OCTRI supports faculty and fellows in establishing and enhancing their careers in clinical and translational research. Explore below for academic educational opportunities, seminars, workshops, and career development funding opportunities.
Human Investigations Program
An integrated, academic curriculum in clinical and translational education, featuring a Master of Clinical Research degree, Certificate in Human Investigations, and individual courses. Review options on the HIP web page.
OCTRI KL2 Program
A two-year career development award for early-career faculty pursuing translational science. Visit the KL2 Program web page.
OSLER TL1 Program
Research experience and mentorship through stipend-funded training. Learn more on the TL1 Program web page.
OCTRI Scholars Program
Resources for promising early-career faculty applying for a mentored career development award or their first independent research grant. Review resources on the OCTRI Scholars web page.
OCTRI Research Forum
Seminar series highlighting practical topics of interest to clinical and translational researchers. Find upcoming seminars on the OCTRI Research Forum web page.
-
In person, on-demand, and live online training
OCTRI provides in person, on-demand, and live online training for clinical research staff. Review your options on the Training and Education for Clinical Research Staff web page.
Clinical Research Coordinator Coffee Chats

We meet every third Tuesday at noon via WebEx. Share your experiences and connect with fellow coordinators.
If you are interested in facilitating one of these discussions or have a suggestion for a topic, please email Kitt Swartz.
Other Training Opportunities

-
OHSU Mentorship Academy

OCTRI has partnered with the OHSU education mission to deliver the OHSU Mentorship Academy, which provides mentors with an intellectual framework, an opportunity to experiment with various methods, and a forum in which to solve mentoring dilemmas with the help of their peers. Find details on the OHSU Mentorship Academy website.
Other resources
Visit the University of Wisconsin Institute for Clinical and Translational Research website for training curricula to further develop your skills.
-
Innovation and entrepreneurship education

OCTRI strives to cultivate innovation in research. Whether you have a burgeoning idea or a proof of concept that is ready for design, OCTRI has education opportunities to meet your needs.
Review opportunities on the Innovation and Entrepreneurship web page.
Featured events
Responsible Conduct of Research

OCTRI is offering an 8-hour seminar series starting February 7 that meets Responsible Conduct of Research requirements for K, T, and other career development awards. Review schedule and details on the event page.

Contact an OCTRI Navigator to connect with research resources

Connect with other coordinators!
Every third Tuesday at noon via WebEx.
Find details on the O2 event page
Questions? Email Kitt Swartz.