First to use “spreading vector” technique
In the quest to cure inherited retinal diseases, Casey Eye Institute is using a novel gene therapy viral vector and surgical approach. The setting? A clinical trial for treatment of X-linked juvenile retinoschisis (XLRS). The sponsor, Atsena Therapeutics, chose Casey Eye Institute as one of just four treatment sites for this clinical trial, and Casey was the first in the world to deploy this innovative therapeutic approach in humans.
Paul Yang, M.D., Ph.D., Martha and Eddie Peterson Endowed Professor and head of the Paul H. Casey Ophthalmic Genetics Division at Casey Eye Institute, says, “We were selected as a clinical site for this study based on our surgical expertise as well as our longstanding record of successful gene therapy trial administration.”
Mark Pennesi, M.D., Ph.D., Professor of Ophthalmology at Casey Eye Institute and Director, Ophthalmic Genetics at the Retina Foundation, emphasizes that Casey is well suited for this and similar trials. “We have a large patient population, and we see people from infancy up to 100 years of age,” he says. “Because genetic diseases are lifelong, we also follow patients for many years.”
Previously, two XLRS clinical trials utilized clinic-based intravitreal injections to deliver a therapeutic agent. Unfortunately, these did not succeed. In contrast, patients in the new Atsena trial receive a surgical pars plana vitrectomy and subretinal gene therapy injection in the operating room. This study also represents the first time a novel “spreading vector” has been used to deliver gene therapy to human patients. This is especially important because of the type of retinal pathology seen in XLRS patients
About XLRS
X-linked retinoschisis is caused by a mutation in the RS1 gene, which encodes the protein retinoschisin. “This protein is involved in intercellular adhesion of the retinal cells, and likely in retinal cell organization and intercellular matrix architecture,” explains Lesley Everett, M.D., Ph.D., M.Phil., Assistant Professor of Ophthalmology and principal investigator for the Atsena study at Casey.
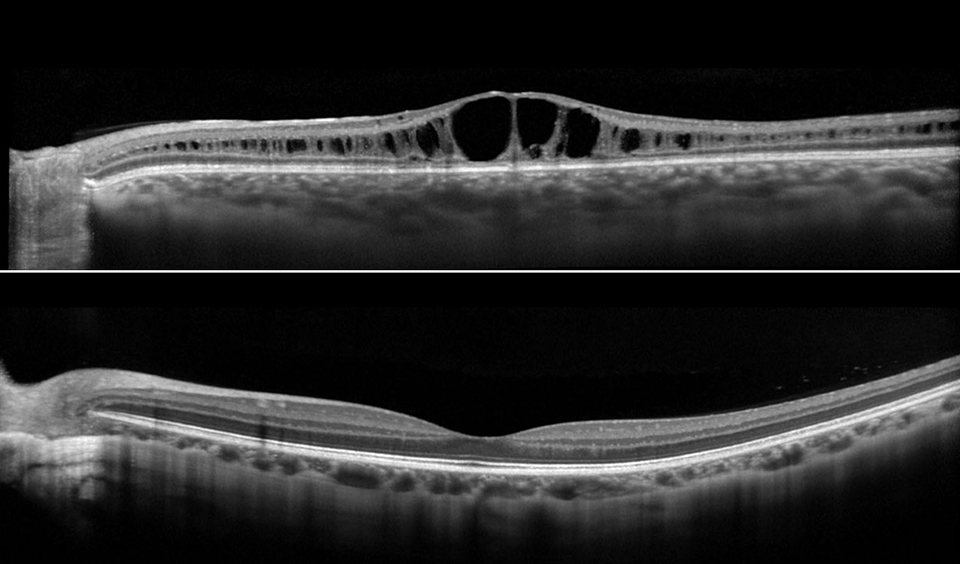
Retinoschisin helps maintain the structural organization of retinal cell layers and promotes visual signal transduction. Eyes with XLRS have splitting of the retinal layers, which leads to photoreceptor degeneration and ultimately, retinal thinning and atrophy.
XLRS is one of the main causes of juvenile macular dystrophy in males, with a prevalence of 1 in every 15,000 to 35,000. The condition was first reported in 1939, but the underlying gene was not identified and cloned until 1997.
The Current Atsena Trial for XLRS
A Phase 1/2, Open-Label, Dose Escalation and Dose Expansion Study to Evaluate the Safety and Tolerability of ATSN-201 Gene Therapy in Male Subjects with RS1-Associated X-linked Retinoschisis (LIGHTHOUSE) (NCT05878860).
How XLRS affects patients
Patients with XLRS typically have symmetric bilateral macular involvement beginning in the first to second decade of life. “It’s often found after a routine vision check at school reveals decreased vision for the first time,” says Dr. Everett. However, the age of onset of signs and symptoms, as well as the severity of vision loss, are quite variable. After childhood, vision often stabilizes until midlife, when degeneration accelerates and, in some cases, leads to legal blindness.
Due to the abnormal retinal anatomy, between 5% and 20% of patients have severe vision loss secondary to retinal detachment. Vitreous hemorrhage is another common complication, especially with peripheral schisis, and may result in deprivational amblyopia in young children. Patients with XLRS should avoid contact sports and ball sports and wear safety glasses due to the risks of retinal detachment and vitreous hemorrhage.
An exciting new direction
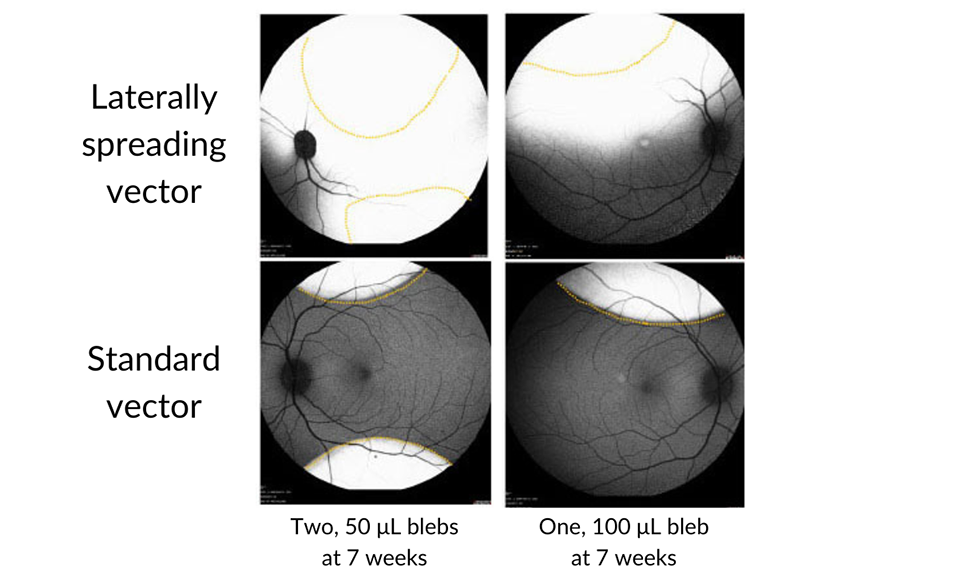
Image modified from original publication: Investigative Ophthalmology & Visual Science June 2022, Vol.63, 2825 – A0341, https://atsenatx.com/our-approach/laterally-spreading-aav.
The previous, unsuccessful XLRS treatment trials used intravitreal injection, in hopes of avoiding the potential surgical risks of retinal detachment or complication due to the abnormal retinal structures in patients with XLRS. The approach used to deliver the Atsena therapy, pars plana vitrectomy and subretinal gene therapy, is now widely used for gene therapy delivery.
“The surgeon uses intraoperative optical coherence tomography (OCT) to create subretinal fluid blebs and uses a fine needle to inject the gene therapy solution under the retina – in this case, out towards the vascular arcades to avoid detachment of the central part of the macula,” says Andreas K. Lauer, M.D., Director of Casey Eye Institute, who performs the surgery.
“The technique requires outstanding dexterity and experience, especially given the fragile structure of retinas affected by XLRS,” says Dr. Everett.
Leading the way in gene therapy
Performing the trial’s first treatment drew on Casey’s depth and breadth of resources. The facility’s large team includes three physician scientists, two genetic counselors, nine clinical research staff members and a large group of technicians who carry out diagnostic testing studies and other facets of the study protocol.
The potential benefits to patients in the trial extends beyond personal health to helping others with retinal diseases. However, the potential for personal benefit is enormous with XLRS.
“This disease affects vision in youth and prime early adulthood,” Dr. Everett says. “Slowing or preventing it could help to prevent a lifetime’s worth of vision loss and the loss of social and educational opportunities for these patients and families.”
Casey’s participation in the XLRS gene therapy trial is also a step forward for genetic medicine. With a history of firsts, including the first patient treated with CRISPR gene editing in the retina, Casey Eye Institute is at the forefront of exploring diverse therapeutic avenues to treat blinding inherited retinal diseases. Each trial adds layers of experience and expertise, expanding the institution's ability to handle complex retinal pathologies and utilize various molecular gene therapy techniques.
Doing what has “always been impossible”
A comment from the trial’s first patient reflects this leap forward in surgical and medical innovation. When Casey doctors and staff thanked the patient for traveling to take part in the trial, he responded, “For me, it’s nothing. You are doing the thing that’s always been impossible” – treating XLRS. Today, he considers the eye treated in the trial to be his stronger eye. This trial, and others like it, are helping Casey Eye Institute open doors to sustained independence and quality of life through preserving vision for patients with rare blinding eye diseases.
Previous XLRS Clinical Trials
In 2015, two human XLRS gene therapy trials were initiated, sponsored by the National Eye Institute (NCT02317887) and Applied Genetics Technology Corporation (NCT0241662).
The NEI trial evaluated the safety of a gene transfer vector (AVV8-RS1) in humans. It found closure of schisis cavities in one individual at higher dosing, which also caused ocular inflammation. Dose-related inflammation was noted.
The AGTC trial sought to evaluate the safety and efficacy of a recombinant adeno-associated virus vector expressing retinoschisin (rAAV2tYF-CB-hRS1) in patients with X-linked retinoschisis. The study was halted due to inflammation and a lack of positive clinical treatment signs.