Translational Examination of Alcohol-Associated Epigenetic Signatures: From Primates to Rodents - Project P003
Project Lead: Verginia Cuzon-Carlson
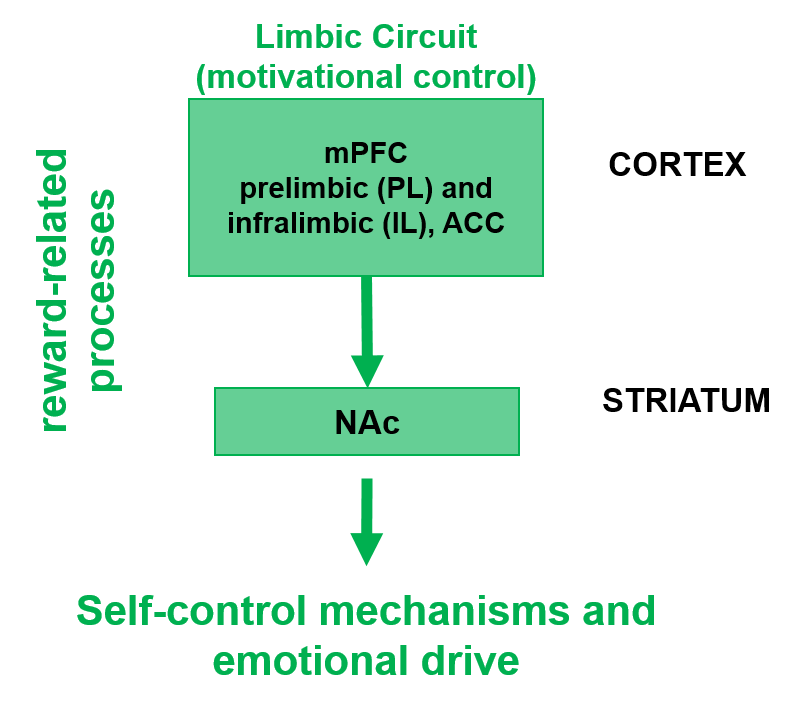
Alcohol use disorder (AUD) is the third leading preventable cause of death in the US. AUD is associated with compulsive heavy alcohol drinking. Currently, there are four FDA approved AUD drugs with varying individual efficacy, resulting in a 66% rate of relapse within months of treatment. Identifying the molecular mechanisms that lead to and maintain risky alcohol drinking behaviors is imperative for the effective design of AUD treatments. Our previous studies illustrate the power of a translational approach that integrates epigenetic, transcriptomic, circuitry, pharmacology and behavioral tools to elucidate the molecular underpinnings contributing to hazardous alcohol use and reduce risky alcohol intake. Using genome-wide DNA methylation (GW-DNAm) analysis of the nucleus accumbens core (NAcC) from low and heavy alcohol-drinking rhesus macaques, we identified differential DNAm (D-DNAm) signals that mapped to genes highly relevant to the tetrapartite synapse that includes pre-and postsynaptic elements, astrocytes, and extracellular matrix, thus impacting the balance between excitatory and inhibitory neurotransmission. Here, we further our understanding of the differential effect of low and heavy alcohol use on the methylome of the limbic cortico-striatal circuit and how those alterations contribute to alcohol-dose dependent adaptations in neurocircuitry and alcohol-drinking behavior. We hypothesize that, in addition to region-specific effects of alcohol, some of the D-DNAm signals previously identified in the NAcC result from cortical inputs into the NAcC in response to alcohol use. Thus, to identify circuit-specific D-DNAm signals, we will compare GW-DNAm datasets from the dorsal anterior cingulate cortex (Brodmann area 32; A32) and NAcC from low and heavy drinking rhesus macaques that consumed alcohol for up to 12 months. A32 was chosen due to its functional and direct anatomical connections with the NAcC. By identifying circuit-specific D-DNAm signals in A32 and NAcC, we will unravel the coordinated epigenomic regulation underlying circuitry in connected brain regions. After integrating DNAm and RNAseq data generated from the same regions and subjects, a subset of compelling, novel gene targets and regulatory regions will be selected and their cell-type of origin will be investigated by conducting amplicon bisulfite sequencing (ABS) in neuronal fractions isolated from the same rhesus brain regions. We will confirm conservation across species by targeted ABS in the prelimbic cortex (PL; rodent equivalent of the primate A32) and the NAcC from a high ethanol-preferring (HP) mouse line that has consumed ethanol for three months. Gene targets that are highly correlated with ethanol dose, conserved across species, and have a proposed functional role in the tetrapartite synapse, will be selected for a high-throughput mouse study using virally-administered genetic manipulation approaches in HP mice. The effects of target manipulation on ethanol consumption and neurotransmission within the A32/PL to NAcC circuit will be evaluated.