Ethanol Effects on Brain Regional Extracellular Matrix Molecules and Cognitive Function - Project P004
Project Lead: Kathy Grant
Co-Investigator: Lawrence Sherman
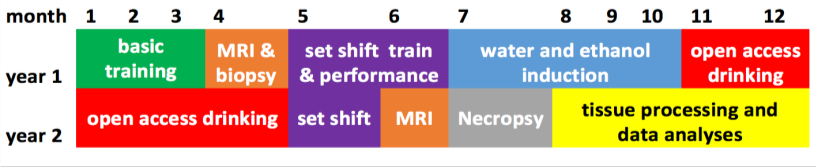
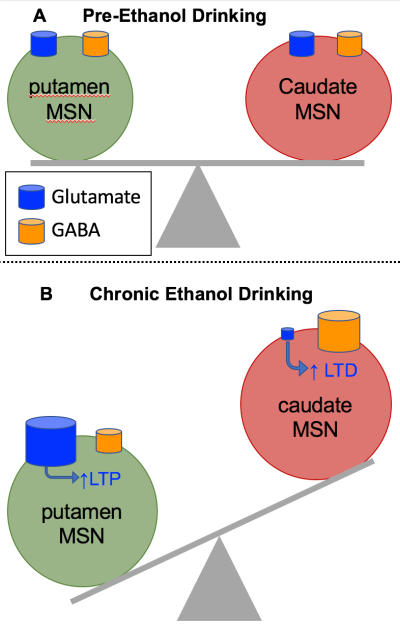
Recent findings from the PARC have identified alcohol (ethanol)-induced alterations in the expression of extracellular matrix (ECM) genes that are associated with our mouse and monkey models of excessive ethanol drinking. In particular, the ECM of perineuronal nets (PNNs) and in synaptic interstitial space appear vulnerable to heavy ethanol drinking, resulting in changes in neuronal activity. In this project, we focus on the ECM glycosaminoglycan hyaluronic acid (HA). HA is a prominent component of PNNs that, in part, help stabilize synaptic contacts, particularly of excitatory glutamatergic (glut) transmission. HA catabolism by hyaluronidases (HYALs) and HA depolymerizing proteins has been linked to alterations in long-term potentiation (LTP), glutamatergic receptors, and neuronal maturation. Our preliminary data and published findings support the hypothesis that ethanol alters the expression of hyaluronidases in the brain and that altered HA catabolism in PNNs of heavy drinkers contributes to the effects of ethanol on neuronal activity. Our focus in this application is on HA integrity of the corticostriatal synapse that may play a role in synaptic plasticity. Thalamo-cortico-striatal loops are organized anatomically to subserve behavioral output ranging from cognitive (flexible, caudate nucleus of the striatum) to highly sensory driven (habitual, putamen region of the striatum) processes. Recordings of medium spiny neurons (MSNs), the main striatal projection neuron, from ethanol drinking monkeys show the balance of Glut and gamma-amino-butyric acid (GABA) mediated synaptic transmission is regionally specific, with greater putamen excitatory tone. These findings suggest that ethanol-induced increased catabolism of HA contributes to the synaptic plasticity in striatal function related to habitual ethanol drinking. In preliminary data from rhesus monkeys, we found strong associations of associative cortical connectivity with flexible cognitive performance and associations of sensorimotor connectivity with risk for chronic heavy drinking. The imaging and cognitive functional measures provide a framework to investigate the role of HA catabolism in risk for, as well as consequence of, chronic ethanol drinking. The timeline for the monkey testing protocol is shown here.