Nerve Fluorophores
Nerve-Specific Fluorophores for Image-Guided Surgery
Significance of Nerve Damage Research
Nerve damage is a significant morbidity associated with surgical intervention. Surgery is among the most commonly recommended treatments for acute and chronic disease with up to 40 million procedures conducted in the United States alone annually. A common, but under recognized side effect of surgical nerve damage is chronic pain following healing of the surgical site. Nerve damage during surgical intervention can also result in loss of function as in the case of nerve-sparing radical prostatectomy, where the functional nerve structures are difficult to visualize. Although neuroanatomy is well known, nerve visualization during surgery continues to challenge even the most experienced clinicians. This difficulty stems from the nature of nerve tissues, which are small, translucent structures that are typically protected deep within the muscle.
Visualization of Nerve Damage in the Operating Room
To aid in nerve visualization in the operating room, a nerve-specific fluorescent contrast agent, specifically one with near-infrared (NIR) absorption and emission would be of immediate benefit to both patients and surgeons. There are a few promising classes of small molecule fluorophores that are known to cross the blood nerve barrier and accumulate in the nerve tissue providing nerve-specific fluorescence signal following systemic administration. However, none of the current fluorophore classes, which include distyrylbenzene (DSB) fluorophores, styryl pyridinium (FM) fluorophores, carbocyanine derivatives, and oxazine fluorophores, are optimized for clinical use.
Nerve-Specific Fluorophores & Administration Methods for Clinical Translation
Research in the Gibbs laboratory is focused on chemical optimization of nerve-specific fluorophores to develop a NIR nerve-specific contrast agent for clinical translation. Translation of fluorescent contrast has lagged behind image-guided surgery system development because each fluorophore is a new molecular entity, which can only be used clinically through an FDA approved investigational new drug (IND) application. IND applications require significant monetary investment in pre-clinical pharmacology and toxicology testing to ensure the new agent is safe for humans. However, this is a financial challenge for fluorophores that will be used only once per patient. To alleviate this difficulty, the FDA approved a new route to the clinic termed an exploratory IND (eIND) in 2006. Under these guidelines, 100 micrograms of a small molecule new molecular entity, such as our nerve-specific fluorophores, can be translated to the clinic without the same costly pre-clinical pharmacology and toxicology testing due to the extremely low dose. Our lab has been developing direct/topical administration strategies for nerve-specific fluorophores that will permit first in human testing of microdoses of our most promising agents facilitating translation to the clinic through an eIND. This collaborative project between the Gibbs Lab, Dr. Jonathan Sorger at Intuitive Surgical and Dr. Adam Alani's Lab at Oregon State University aims to move nerve-specific fluorophores to the clinic to guide oncologic surgeries such as nerve-sparing prostatectomy and orthopedic limb repair surgeries.
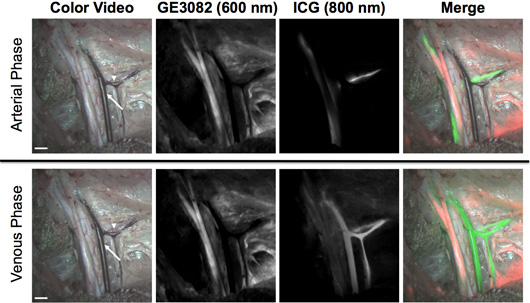
Distyrylbenzene Fluorophores for Simultaneous Nerve and Vessel Imaging
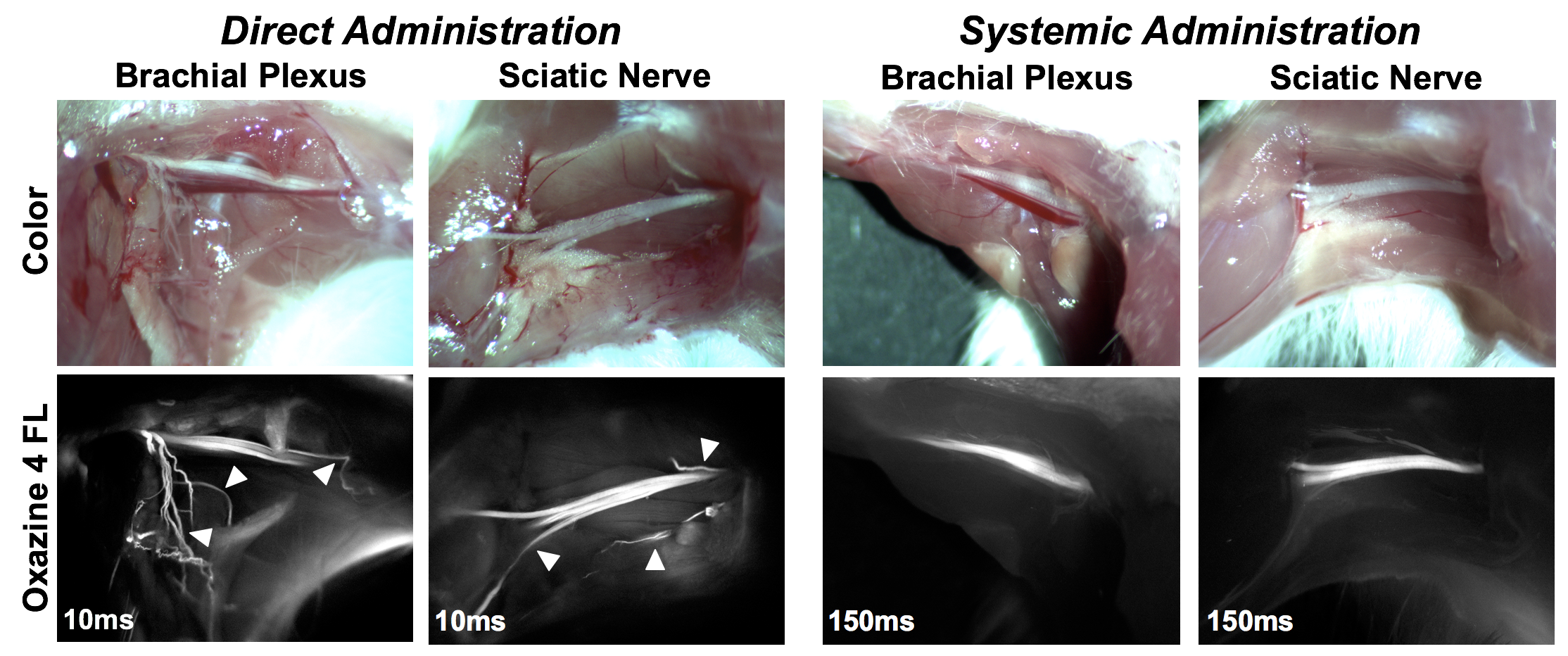
Direct administration strategy
Acknowledgments
This research is funded by the National Institute of Biomedical Imaging and Bioengineering (NIBIB) and the Oregon Clinical & Translational Research Institute (OCTRI).