ES Cell Derived Chimeras Background
Background
Introduction of mutant ES cells into developing embryos is an alternative way to generate novel genetically modified mouse lines. The transfer of mutant ES cells into pre-implanted embryos and subsequent transplantation of chimeric embryos in the uterus of a pseudopregnant female does not affect their development which leads to the birth of a normal viable progeny. During embryogenesis, ES cells can colonize all tissues of the chimeric embryo, including the germ cell lineage (germline) when the differentiated ES cells become involved in spermatogenesis of the adult chimeras. The resulting chimeras will transmit mutation to their offspring and as a result novel mutant mouse lines will be produced. Hence, ES cells serve as a bridge between in vitro genetic manipulations and the in vivo consequences of these manipulations. One of main requirements for a successful application of the gene targeting technology is the capacity of ES cells to give origin for gametes (germline formation) in a chimeric organism. It is known that long term culture of ES cells leads to the block of germline transmission (Nagy et al., 1993;Fedorov et al., 1997) that apparently results from accumulation of spontaneous genetic or epigenetic changes of the ES cell genome. Therefore it is necessarily to keep ES cells under optimal cell culture conditions and to perform a minimal number of passages.
Various methods were established to introduce ES cells into host embryos, these include: injection into the blastocyst cavity (Moustafa, Brinster, 1972), injection under the zona pellucida of morulas (Lallemand, Brûlet, 1990) and aggregation with a morula (Wood et al., 1993). Injecting into the blastocyst cavity is now the most commonly used approach for the production of ES cell chimeras.
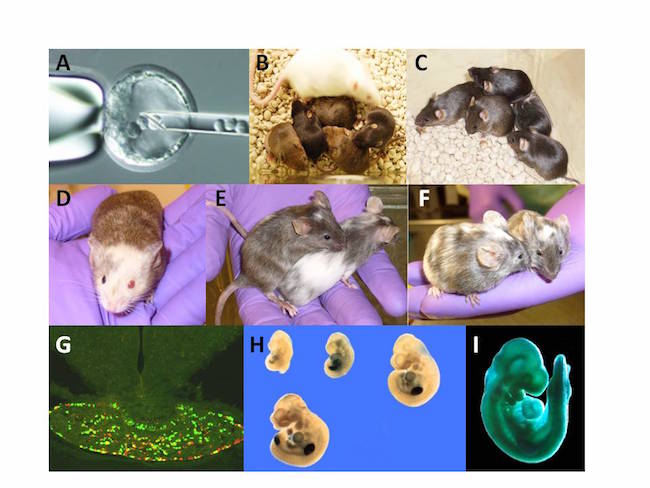
Generation and analysis of chimeras. (A) Injection of ES cells into a blastocyst. (B, C, D) Chimeric mice derived from mutant ES clones with the genetic background of 129/Sv, C57Bl/6 agouti and 129/Ola respectively. Chimeras (B, C and D were produced by injection ES cells (129/Sv background) into C57BL/6 B6 blastocysts for Dr. J. Christian (University of Utah) and Dr. A Barnes (OHSU, Portland, Or.). Respectively all chimeras demonstrated high intensity of agouti coloration of the fur and further germline transmission. Chimeras (B,C,D,E) were produced by the OHSU TMM within 2009-2012. (E, F) Chimeric mice were produced by injection of ES clones (C57Bl/6) into C57BL/6-Albino blastocysts for Dr. E. Tucker (West Virginia University School of Medicine) and Dr. P.G. Barr-Gillespie (OHSU, Portland, Or).(G) Expression of green fluorescent and red fluorescent reporter genes in distinct subpopulations of cells in the pituitary of a POMC/GFP-POMC/RFP aggregation chimeric mouse produced for Dr. M.J. Low (University of Michigan). (H) The limb specific expression of the beta-gal reporter gene in gene trap embryos at E9, E9.5, E10.5 (upper row, left to right), and E11.5 (lower row), respectively. (I) Ubiquitous expression of the b-gal in gene trap embryos at E9.5 (Project of TMM, OHSU).