Projects
Activity-dependent plasticity in the entorhinal-dentate gyrus
Our lab has focused for a number of years on the role of adult-generated granule cells in the dentate gyrus with an emphasis, not on neurogenesis per se, but rather on the integration of these newly generated granule cells into functioning circuits in the dentate gyrus. The rationale for this work is that a substantial proportion of these new cells (4000 per day in a mouse) survive and mature into fully functional granule cells, but have a specific function in the formation of new memories. Our earlier work, using both a novel mouse model (POMC-EGFP), as well as viral-mediated labeling with retroviral and lentiviral vectors in vivo, allowed us to define the stages of integration and probe the role of specific molecules such as PTEN and miR128. These studies provided evidence of discrete time periods in which new neurons establish connections and have distinct inputs. For example, our recent data show that lateral perforant path inputs from lateral entorinal cortex provide preferential synaptic input to newly-integrated granule cells. This provides a unique model system to examine activity-dependent reshaping of the neuronal circuit in an adult environment. We are exploring these questions using both physiological and molecular approaches.
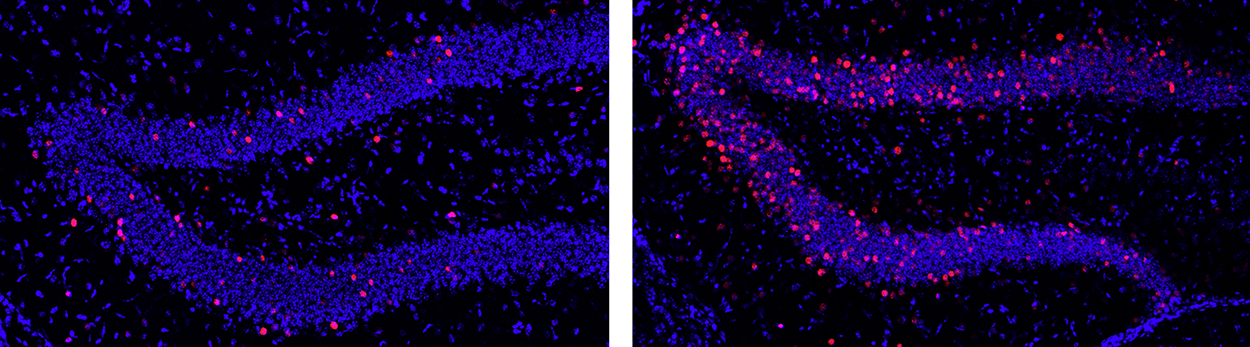
Figure 1 Dentate granule cells in the mouse hippocampus were permanently labeled after a single 2-hour period of wheel running (right) compared to the very low level of neuronal cell activity in the resting home cage environment (left). Activated cells appear as red dots. Exercise occurred 3 days prior to these images. These cells are being collected by laser capture and analyzed for changes in gene expression (Chatzi, unpublished).
In collaboration with the lab of Richard Goodman we wanted to look at cell- and time-specific gene expression in newly-generated as well as mature granule cells in the dentate gyrus. To examine activity-dependent gene expression, we used a discrete physiological stimulus — a single two hour exposure to voluntary exercise on running wheels, akin to a single trip to the gym. This approach allows for precise temporal control of the stimulus (and the response). We took advantage of the “sparse” nature of the dentate granule cell network in which very few cells are active under baseline conditions. We used the cFoscre/ERT2/Luo mouse, created by Liqun Luo, to permanently mark a cohort of granule cells that were activated by the stimulus in a discrete time window as defined by a tamoxifen injection. We are most interested in genes that reshape synaptic or circuit connections after a transient stimulus — the equivalent of an impulse function in engineering terms. We found a significant increase of the cells marked as activated during the 2-hour exercise period. We isolated the “activated” cells using laser capture microdissection, and RNA from microdissected cells was reverse transcribed and processed for RNASeq. This analysis has revealed a number of interesting candidate genes. Experiments are now focused on establishing the relevance of these genes to morphological and functional measures of synapse formation and activity. On a physiological level, a striking effect is that the exercise increased dendritic spines and synaptic activity in a laminar-specific manner — i.e. only in the outer molecular layer, which receive inputs from the lateral entorhinal cortex and convey specific “contextual” information. We would like to know how this occurs, how rebalancing on inputs onto newly-integrated granule cells occurs and whether it is exercise-specific.
Most recent publication from this project
Exercise-induced enhancement of synaptic function triggered by the inverse BAR protein, Mtss1L
Chatzi C, Zhang G, Hendricks WD, Chen Y, Schnell E, Goodman RH*, Westbrook GL*
Elife 2019 Jun 24; 8:e45920 *co-senior authors
Probing anti-NMDA receptor encephalitis with a novel mouse model
Neurologists have long recognized a syndrome of “limbic encephalitis” characterized by behavioral changes, psychosis, memory loss and seizures accompanied by increased MRI signal in the hippocampus and limbic system. The underlying cause of many of these cases went unrecognized until about 10 years ago when Dalmau and colleagues reported the presence of anti-NMDA receptor antibodies in the serum of a number of such cases. Subsequent papers have characterized the clinical characteristics, and treatment approaches to remove the antibodies (by plasmapheresis) or suppress the immune system (with non-specific drugs) have led to recovery in some cases. However, recovery is often incomplete and prolonged. Once recognized, this disease is surprisingly common, can occur in children as well as adults, and can be associated with permanent disability or death. This disorder has received substantial attention in the lay press including a patient account (Brain on Fire, Susan Cahalan). The idea of autoimmunity against brain and spinal cord proteins is not new of course and is the cause of demyelinating diseases such as multiple sclerosis as well as myasthenia gravis (antibodies to AChRs and related proteins). However many questions remain poorly understood for anti-NMDA receptor encephalitis concerning the trigger for the autoimmune reaction, the epitopes involved, the role of B vs. T cell-mediated immunity in the pathophysiology, whether anti-NMDA receptors antibodies are function-blocking or trigger endocytosis, and whether the clinical symptoms are primarily a result of NMDA receptor hypofunction or the accompanying neuroinflammation.
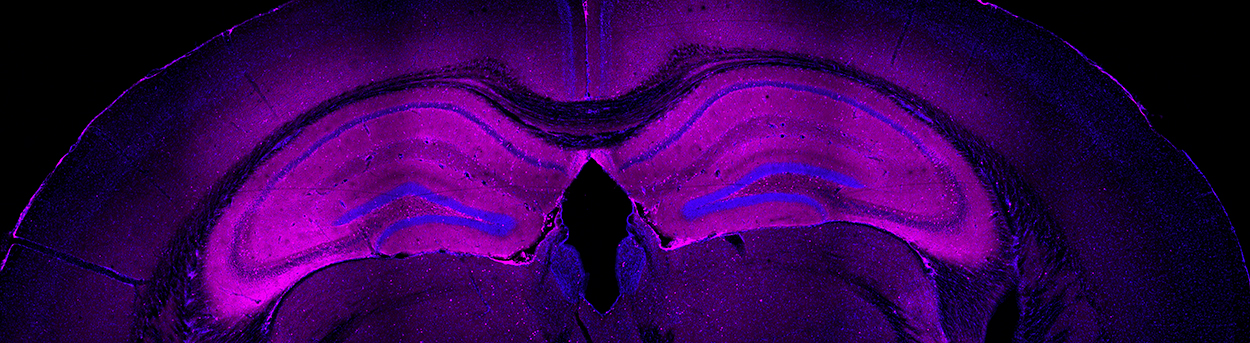
Figure 2 IgG infiltration of adult mouse brain following immunization with holo-NMDA receptors in proteoliposomes. Note intense staining (pink) of hippocampus. Liposome control had no IgG staining (not shown) (Jones, unpublished).
No animal model exists that would allow such questions to be directly addressed. Thus we decided, in collaboration with the Gouaux lab at the Vollum Institute, to develop a rodent model to see if we could test key questions in this disorder. In the first set of experiments, adult mice were injected with NR1/NR2B holomeric proteins in proteoliposomes, given a booster at 2 weeks and examined at 6–12 weeks. The mice developed a characteristic syndrome of locomotor and behavioral changes, seizures and increased mortality. Histology and immunohistochemistry revealed inflammatory infiltrates characteristic of encephalitis, particularly prominent in the hippocampus (Figure 2). There were T cells as well as B cells in brain tissue. Analysis of serum in heterologous cells expressing NMDA receptors and by Western blot confirmed the presence of NMDA receptor antibodies. Thus we are now in position to ask a number of key questions regarding the pathophysiology and potentially test treatment strategies.
Most recent publication from this project
Autoimmune receptor encephalitis in mice induced by active immunization with conformationally stabilized holoreceptors
Jones BE, Tovar KR, Goehring A, Jalali-Yazdi F, Okada NJ, Gouaux E, Westbrook GL
Science Translational Medicine 2019 July 10; 11(500):eaaw0044