Projects
Determine the mechanism of pregnancy-associated breast cancer
We found that a breast cancer diagnosis within 10 years of a childbirth carries a ~3 to 5-fold increased risk of developing metastatic cancer compared to breast cancer diagnosed during pregnancy or in women who have never been pregnant. The cause of the poor prognosis of postpartum breast cancer is a major area of research. Importantly, postpartum breast cancer impacts ~50% of all young breast cancer patients. In collaboration with Dr. Virginia Borges (University of Colorado, Anschutz Medical Campus), we established a multidisciplinary, translational research program to address the unmet clinical needs of women diagnosed with postpartum breast cancer. The summation of our preclinical and retrospective clinical outcomes studies have identified postpartum mammary gland involution, and its associated wound healing-like stromal milieu, as the principal driver of metastasis. Further, we have identified the efficacy of COX-2 targeted interventions in rodent models of postpartum breast cancer, leading to three phase 0 clinical trials aimed at translating our discoveries to young breast cancer patients. Ongoing preclinical studies in rodent models include investigating the roles of specific immune cell populations modulating postpartum disease progression, and determining the underlying mechanism of NSAID efficacy in the context of the post-weaning, involuting mammary gland.
Distinguishing indolent from life-threatening disease
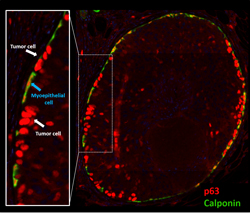
A second research effort is aimed at developing diagnostic tools to distinguish indolent from lethal breast cancers, as current breast cancer screening triggers a spectrum of potentially unnecessary treatments and psychological distress. This is particularly true for women diagnosed with Ductal Carcinoma in Situ. We are investigating the tumor suppressive attributes of the mammary myoepithelial cell using genetically engineered murine models, 3D co-culture models with primary myoepithelial cells, and a human DCIS tissue cohort with available outcomes data.
Identify the ECM proteomes of the 'breast-cancer protected' and 'at-risk' mammary gland
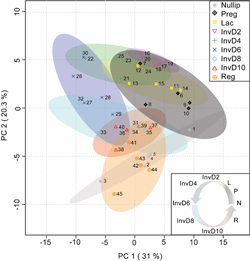
Our lab has been at the forefront of investigating the role of the extracellular matrix in normal and pathologic breast development and has made seminal contributions in defining the functional unit of the mammary gland as the epithelial cell plus its surrounding ECM. In collaboration with Dr. Kirk Hansen (University of Colorado, Anschutz Medical Campus), we have substantially improved mass-spectrometry methods to identify ECM proteins as well as to determine their absolute abundance in complex mixtures. We have developed methods for endogenous tissue matrix isolation from mammary gland and liver, permitting functional assessment including roles in stem cell niches. Ongoing projects include determining fibrillar collagen regulation in the mammary gland to gain insight into breast density and breast cancer metastasis.