OHRC Faculty
Alfred L. Nuttall, Ph.D., Director

Email: nuttall@ohsu.edu
Phone: 503-494-8032
Lab phones:
503-494-2930 (HRC 0435)
503-494-2993 (HRC 0429)
503-494-2937 (HRC 3rd Floor Bay "B")
Office: HRC 0410A-B
Research Interests: cochlear mechanics, cochlear blood flow, optical microangiography, laser vibrometry, molecular mechanisms, signal transduction
Alfred L. Nuttall, Ph.D. is the director of the Oregon Hearing Research Center at OHSU, where he is a Professor in the Department of Otolaryngology-Head and Neck Surgery, the Vice Chairman for Research and the first Jack Vernon Endowed Professor in Hearing Research. Dr. Nuttall joined OHSU in 1996. Additionally, he is a Professor Emeritus of Otolaryngology at the University of Michigan.
Background
Dr. Nuttall received a B.S. degree in electrical engineering from Lowell Technological Institute, Lowell, MA, in 1965, two M.S. degrees in bioengineering and electrical engineering and a Ph.D. degree in bioengineering from the University of Michigan, Ann Arbor, in 1968, 1969, and 1972, respectively. His postdoctoral work was completed at Kresge Hearing Research Institute, Department of Otolaryngology, University of Michigan, where he became an Assistant Professor in 1976, a Professor in 1987, and a Professor-Emeritus in 1996. Thereafter, he joined Oregon Health & Science University, Portland, where he is currently a Professor of Otolaryngology and Director of the Oregon Hearing Research Center, Vice Chair for Research and Jack Vernon Endowed Professor in Hearing Research at Oregon Health & Science University. He is the author or coauthor of over 200 peer-reviewed articles on cochlear mechanics and cochlear blood flow.
Previous Appointments
- Professor, Associate Professor and Assistant Professor in the Department of Otolaryngology at the University of Michigan
Degrees
- B.S. degree in electrical engineering, Lowell Technological Institute (now the University of Massachusetts at Lowell)
- M.S. degrees in bioengineering and electrical engineering, University of Michigan
- Ph.D. degree in bioengineering, University of Michigan
- Postdoctoral work at the Kresge Hearing Research Institute, University of Michigan
Research Interests
- Hearing function and hearing loss
- Cochlear physiology, with particular interest in:
How does load sound cause hearing loss?
How do the sensory cells of the organ of Corti amplify and discriminate complex sounds
Major Milestones and Significant Discoveries
Proof that spontaneous otoacoustic emissions come from vibration of the basilar membrane:
- The cochlea produces nitric oxide (NO) in abundance
- The organ of Corti produces power in response to sound
- A technology to measure human cochlear blood flow
Summary of Research Interests
Dr. Nuttall's research focuses on hearing function and hearing loss. The Nuttall lab is interested in how loud sounds causes hearing loss and how the sensory cells of the organ of Corti amplify and discriminate complex sounds.
Dr. Nuttall's research interests in cochlear physiology are divided into two major areas:
1) Mechanical processing of acoustic energy and function of the sensory cells,
2) The control of inner ear blood flow and the pathophysiology of cochlear blood circulation including the cellular and molecular mechanisms of noise-induced hearing loss in the vascular tissue, lateral wall and in the sensory cells of the organ of Corti.
The measurement of physiological variables for cochlear mechanics and blood flow is accomplished with the use of state of the art approaches. For example, laser interferometry is used to determine the motion of cellular structures in the organ of hearing, the organ of Corti, and Doppler optical microangiography is used to make direct observations of blood flow responses to loud sound in capillaries within the cochlea. Loud sound induced hearing loss elicits a multitude of deleterious responses including hypoxia/ischemia, inflammation, increased metabolic stress and vascular permeability, all which increase reactive oxygen species and may result in cell death. Dr. Nuttall's lab is studying the mechanisms by which loud sound-induced signal transduction pathways which contribute to hearing loss.
In the News
- Oregonian 11-30-2016
- The Business Journals
- OHSU News
Additional information is available on his lab page. To access his publications, please click here to go to PubMed.
Selected Publications
Ramamoorthy, S., Zhang, Y., Petrie, T., Fridberger, A., Ren, T., Wang, R.-K., Jacques, S. and Nuttall, A.L. (2016) Minimally invasive surgical method to detect sound processing in the cochlear apex by optical coherence tomography. J Biomed Opt 21(2), 025003. doi:10.1117/1.JBO.21.2.025003. PMC4796094
Wu, T., Ramamoorthy, S., Wilson, T., Chen, F., Porsov, E., Subhash, H., Foster, S., Zhang, Y., Omelchenko, I., Bateschell, M., Wang, L., Brigande, J., Jiang, Z.-G. and Nuttall, A.L. (2016) Optogenetic control of mouse outer hair cells. Biophys J 110(2):493-502. PMC4724629
Yang, Y., Chen, F., Karasawa, T., Ma, K.-T., Guan, B.-C., Shi, X.-R., Li, H., Steyger, P., Nuttall, A.L. and Jiang, Z.-G. (2015). Diverse Kir expression contributes to distinct bimodal distribution of resting potentials and vasotone responses of arterioles.PLoS ONE, 10(5), e0125266. doi:10.1371/journal.pone.0125266. PMC4418701
Wilson, T., Omelchenko, I., Foster, S. Zhang, Y., Shi, Xiaorui and Nuttall, A.L. JAK2/STAT3 inhibition attenuates noise-induce hearing loss. PLoS One (2014) 9(10):1-10. DOI: 10.1371/journal.pone.0108276. PMC4183445
Le Floc'h, J., Tan, W., Telang, R.S., Vlajkovic, S.M., Nuttall, A.L., Rooney, W.D., Pontre, B. and Thorne, P.R. Markers of cochlear inflammation using MRI. J Magn Reson Imaging (2014) 39(1):150-61. PMC3935384
Ramamoorthy, S., Zha, D., Chen, F., Jacques, S.L., Wang, R., Choudhury, N., Nuttall, A.L. and Fridberger, A. Filtering of acoustic signals within the hearing organ. J Neurosci (2014) 34(27):9051-58. PMC4078082
Shi, X., Zhang, F., Urdang, Z., Dai, M., Neng, L., Zhang, J., Chen, S., Ramamoorthy, S. and Nuttall, A.L. Thin and open vessel-windows for intra-vital fluorescence microscopy imaging of the murine cochlear lateral wall. Hear Res (2014) 313:38-46. PMC4176943
Ren, T., Zheng, J., He, W. and Nuttall, A.L. Measurement of amplitude and delay of stimulus frequency otoacoustic emissions. J Otol (2013) 8(1):57-63. PMC4097125
Ramamoorthy, S. Wilson, T., Wu, T. and Nuttall, A.L. Non-uniform distribution of outer hair cell transmembrane potential induced by extracellular electrical field. Biophys J (2013) 105(12):2666-75. PMC3882456
Reif, R., Zhi, Z., Dziennis, S., Nuttall, A.L. and Wang, R.K. Changes in cochlear blood flow in mice due to loud sound exposure measured with Doppler optical microangiography and laser Doppler flowmetry. Quant Imaging Med Surg (2013) 3(5):235-42. PMC3834207
Han, W.J., Shi, X. and Nuttall, A.L. Noise-induced nitrotyrosine increase and outer hair cell death in the guinea pig cochlea. Chin Med J (2013), 126(15):2923-2927.PMC3947561
Subhash, M.S., Choudhury, N., Chen, F., Wang, R.-K., Jacques, S. and Nuttall, A.L. Depth-resolved absolute vibrometry based on Fourier domain low coherence interferometry. J Biomed Opt (2013) 18(3):036003. doi:10.1117/1.JBO.18.3.036003 PMC3584824
Reif, R., Qin, J., Dziennis, S., Zhi, Z., Nuttall, A.L. and Wang, R.K. Monitoring hypoxia induced changes in cochlear blood flow and hemoglobin concentration using a combined dual-wavelength laser speckle contrast imaging and Doppler optical microangiography system. PLoS One (2012) 7(12):e52041. doi: 10.1371/journal.pone.0052041 PMC3525546
Dziennis, S., Reif, R., Zhi, Z., Nuttall, A.L. and Wang, R.K. Effects of hypoxia on cochlear blood flow in mice evaluated using Doppler optical microangiography. J Biomed Opt (2012) 17(10):106003. doi:10.1117/1.JBO.17.10.106003. PMC3461130
Ramamoorthy, S. and Nuttall, A.L. Half-octave shift in mammalian hearing is an epiphenomenon of the cochlear amplifier. PLoS One (2012) 7(9):e45640. doi: 10.1371/journal.pone. 0045640 PMC3458085
Nuttall, A.L. and Fridberger, A. Instrumentation for studies in cochlear mechanics: From von Békésy forward. Hear Res (2012) 293(1-2):3-11. PMC3483786
Subhash, M.S., Nguyen-Huynh, A., Wang, R.K., Jacques, S.L. and Nuttall, A.L. Feasibility of spectral-domain phase-sensitive optical coherence tomography for middle ear vibrometry. J Biomed Opt (2012) 17(6):060505. doi:10.1117/1.JBO.17.6.060505 PMC3381045
Zha, D., Chen, F., Ramamoorthy, S., Fridberger, A., Choudhury, N., Jacques, S.L., Wang, R.K., and Nuttall, A.L. In vivo outer hair cell length changes expose the active process in the cochlea. PLoS One (2012) 7(4):e32757. PMC3322117
He, W., Porsov, E., Kemp, D., Nuttall, A.L., Ren, T. The group delay and suppression pattern of the cochlear microphonic potential recorded at the round window. PloS One (2012) 7(3):e34356. doi: 10.1371/journal.pone.0034356 PMC3314608
Ramamoorthy, S. and Nuttall, A.L. Outer hair cell somatic electromotility in vivo and power transfer to the organ of Corti. Biophys J (2012) 102:388-398. PMC3274794
See more of Dr. Nuttall's publications at PubMed.
Nuttall Lab

Lab Phone: 503-494-2930 (HRC 0435)
503-494-2993 (HRC 0429)
503-494-2937 (3rd floor bay B)
Mailing address: Alfred Nuttall Lab
Oregon Health & Science University
Oregon Hearing Research Center
Mail Code: NRC04
3181 SW Sam Jackson Park Road
Portland, OR 97239
Current Lab Members
Teresa Wilson
Senior Research Associate
PubMed
George Burwood
Research Instructor
Rubing Xing
Senior Research Assistant
Current Lab Projects
Dr. Nuttall's research specialization is in cochlear physiology with a dual focus on cochlear mechanics and cochlear blood flow (CoBF). In the study of cochlear mechanics, his laboratory currently works on determining the micromechanical motions, forces, and electrochemical signals needed for the "cochlear amplifier." The biological amplifier powers the extraordinary sensitivity of the ear for detection of sound. For the area of cochlear blood flow, his laboratory studies the cellular mechanisms of noise-induced hearing loss in the vascular tissue and in the sensory cells of the organ of Corti. This damage includes a form of stress induced inflammation. The goal of the laboratory is to understand the molecular mechanisms of the damage from exposure to loud sound. An additional goal is to develop imaging technology for the clinical assessment of altered blood flow in the human ear and for the diagnosis of Ménière's disease.
Cochlear Mechanics
The broad goal is to understand the electro-mechanical processing of acoustic energy by the cells of the organ of Corti. This process consists of the transduction of the sound stimulated vibration of the organ through movement of stereocilia of hair cells. This movement gates a mechano-sensitive channel. One type of hair cell, the outer hair cell, has a voltage-sensitive motor capable of changing cell mechanical properties at great speed. These properties of hair cells set the stage for what is thought to be a feedback amplifier of the acoustic stimulus. The pathophysiology of sound is studied by measurements of the dynamic changes in the electrical and mechanical properties of the organ. Pharmacological and genetic approaches are used to manipulate hair cells in combination with sound.
Current Projects
Developing non-invasive techniques to perform structural and vibrational imaging of the microstructure of the hearing organ (organ of Corti). Laser Doppler vibrometry (LDV) is a non-invasive tool for the study of cochlear mechanics. There has been a growing interest in the field of hearing research to adopt optical coherence tomography (OCT) as a tool for the studies of cochlear mechanics. We developed recently, a Fourier domain OCT (PSFDOCT) for vibration of tissue at a sub-nanometer scale with dual angle delay encoded small beam capable of measuring both radial and transverse motion.
Investigating the mechanisms underlying speech-frequency hearing. To understand how the cochlea encodes speech, we need to examine regions that actually represent speech, since several studies indicate that mechanisms of low frequency hearing differ substantially from those in the extensively studied cochlear base, which mediates high-frequency hearing. Sound entering the outer ear vibrates the stapes and stimulates the cochlea via the launch of a fluid-structure traveling wave along the structures in the inner ear. The constituent sound frequencies are separated along the length of the cochlea with high frequencies near the stapes, which inputs sound energy and low frequencies near the cochlear apex. The sound-induced vibrations inside the cochlea are highly complex in a living mammal and are vulnerable to damage. These vibrations could, therefore, provide important information on the mechanisms of normal and impaired hearing. Recent developments, pioneered largely by our group have made it feasi¬ble to study the previously inaccessible low-frequency parts of the cochlea in vivo, without creating an artificial opening in the bony capsule. These advances remove concerns about artifacts that plagued research on low-frequency hearing mechanisms for decades and we are now entering a new era of minimally invasive in vivo studies. These studies will help us understand how the cochlea encodes speech, and may allow optimized stimulation paradigms to be designed for cochlear implants.
Cochlear Blood Flow
The overarching goal of these studies is to learn the role of inner ear blood flow in normal cochlear function (homeostasis) and pathological conditions such as loud sound induced damage. The blood flow to the inner ear is supplied by a single end artery, the spiral modiolar artery. This small artery has various mechanisms of vasoconstriction and dilation. The capillary-based control of flow and vascular permeability of the capillaries are being studied as a pathological mechanisms of age and noise induced hearing loss. Current projects: In collaboration with Drs. Ricky Wang and Timothy Hullar, we are currently developing an outpatient optical coherence tomography/optical microangiography (OCT/OMAG) instrument capable of taking 3D images of the cochlea. The goal of this translational research is to use OMAG in clinical settings for quantitatively measuring CoBF and imaging Reissner’s membrane in order to aid in the diagnosis of SSHL and Meniere’s disease. With this instrument, we will be able to measure CoBF and, ultimately, to categorize the flow level as normal or abnormal and to identify the position of the Reissner’s membrane. This information can reveal whether cochlea hydrops is present and, in turn, can assist in the diagnosis of Meniere’s disease. Our instrument will also provide data that will be useful in determining metrics for a diagnosis of ‘vascular’ sudden sensorineural hearing loss (SSHL) and in developing a rational treatment for SSHL, involving vasoactive and anti-inflammatory agents.
Sound Induced Signal Transduction Mechanisms Leading to Cellular Pathology
Current projects focus on the signal transduction mechanisms by which loud sound alters blood flow, vascular permeability and cellular damage in multiple cells types in the cochlea including outer hair cells, fibrocytes and endothelial cells. We have found that the JAK2/STAT3 plays a key role in the generation of reactive oxygen species in outer hair cells. Signal transducer and activator of transcription 3 (STAT3) is a transcription factor, activated by phosphorylation in response to cytokines, hormones, growth factors and vasoactive agents. Transcriptional targets of STAT3 include vascular endothelial growth factor, manganese superoxide dismutase and survivin. Inhibiting the JAK2/STAT3 pathway (with JSI-124) reduces loud sound-induced reactive oxygen species in outer hair cells as shown by fluorescent intensity (CellROX Green, 488) at 2 h following noise exposure (NE) shown below. Further studies reveal that STAT3 inhibition increase outer hair cell survival within the basal region of the cochlea improves auditory brainstem responses.
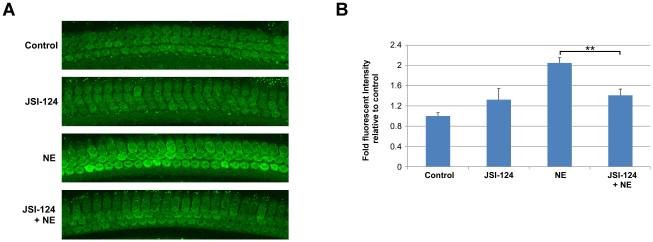
We are also investigating the mechanisms by which loud sound induced temporary hearing loss induced hypoxia/ischemia can result in dynamic changes in endothelial cell cytoskeletal organization, a hallmark of vascular permeability change and loss of barrier function in the stria vascularis. VE-Cadherin is required for maintaining a restrictive endothelial barrier. We have been able to image endothelial cells of intact isolated capillaries in the stria vascularis with super resolution microscopy, and are investigating upstream signaling cascades leading to the alterations in VE-cadherin location and endothelial cell function.
Collaborators
Anders Fridberger, Ph.D.
Professor of Neuroscience Linkoping University
Linkoping, Sweden
Ruikang Wang, Ph.D.
Professor, Bioengineering
University of Washington-Seattle
Additional Information
Timothy Hullar, M.D.
Professor, Otolaryngology/Head and Neck Surgery
Oregon Health & Science University
Steve Jacques, Ph.D.
Professor, Departments of Biomedical Engineering and Dermatology
Lina Reiss, Ph.D.
Assistant Professor, Oregon Hearing Research Center
Tianying Ren, Ph.D.
Professor, Oregon Hearing Research Center
Peter Barr-Gillespie, Ph.D.

Email: gillespp@ohsu.edu
Phone: 503-494-2936
Lab phone: 503-494-2950
Office: MRB 0920
Research Interests: hair cells, mechanotransduction, myosin, motors, ion channels, cytoskeleton, pumps and transporters, inner ear, hearing, balance, molecular
Peter G. Barr-Gillespie, Ph.D., is a Professor with the Oregon Hearing Research Center and an Affiliated Scientist with the Vollum Institute. He has been with OHSU since 1999.
Current Appointments
- Chief Research Officer and Executive Vice President
- Professor in the departments of Otolaryngology-Head and Neck Surgery, Cell Biology & Development and Biochemistry & Molecular Biology
- Senior Scientist, Vollum Institute
- Previous appointments:
- Assistant Professor and Associate Professor of Physiology, Johns Hopkins University
Degrees
- B.S. degree in chemistry, Reed College
- Ph.D. degree in pharmacology, University of Washington
- Postdoctoral work at UCSF, UT Southwestern Medical Center
Research Interests
- Hair-cell transduction
- Hair-bundle development
Major Milestones & Significant Discoveries
- Development of methods for isolation of hair bundles and analysis of constituent proteins and lipids via mass spectrometry
- Determination of roles for myosin Ic, myosin VI, and myosin VIIA in adaptation and bundle structure
- Characterization of the structure, identity, and regeneration of the tip link
- Description of homeostatic mechanisms used by hair bundle to handle Ca2+ (Ca2+ pump, ATP delivery, H+ transporter)
- Elucidation of protein expression and localization steps required for assembly of the hair bundle
More about Professor Barr-Gillespie
See Peter Barr-Gillespie's faculty webpage at The Vollum and his publications at PubMed.
Barr-Gillespie Lab
Please access Professor Barr-Gillespie's lab page here.
John V. Brigande, Ph.D.

Email: brigande@ohsu.edu
Lab phone: 503-494-4568
Office: HRC 0414
Research Interests: retroviral lineage analysis, fate map, fetal gene transfer, fetal pharmacotherapy, gene therapy for congenital deafness
John V. Brigande, Ph.D. is a Professor at the Oregon Hearing Research Center.
Current Appointments
- Professor of Otolaryngology
- Professor of Cell, Developmental, and Cancer Biology
Background
John V. Brigande's introduction to neuroscience formally began in Thomas N. Seyfried's lab at Boston College where his Master's work focused on reactive astrocytosis in a mouse model of human temporal lobe epilepsy. Brigande then transitioned to developmental neurobiology for his doctoral work again with Dr. Seyfried studying glycosphinogolipid biosynthesis in the organogenesis-stage mouse embryo. His doctoral studies ignited a passion for mammalian development that was fostered by postdoctoral training in developmental genetics with Karen Artzt at the University of Texas at Austin and in auditory development with Donna M. Fekete at Purdue University. Brigande began his assistant professorship in the Oregon Hearing Research Center in July 2003 and was promoted to associate professor in 2009. Brigande joined the Hearing Health Foundation's Hearing Restoration Project in 2011 and works on the definition of mouse model systems to test candidate genes for hair cell regeneration.
Summary of Current Research
We study the development of the mammalian inner ear using mouse as a model system. Our approach involves experimental embryology, a palette of surgical, micromanipulation, and gene transfer techniques that enables interrogation of developmental processes in vivo. Our long term goal is to apply what we learn about the genetic mechanisms governing inner ear sensory organ development to define and validate efficacious gene and cell therapies to treat hearing loss and balance disorders.
Retroviral lineage analysis enables us to understand the types of cell fate choices an otic progenitor makes and the timing of those choices during sensory organ formation. We have shown by transuterine microinjection of lineage virus at embryonic day 11.5 (E11.5) that both auditory and vestibular hair cells are lineally related to their supporting cells and that spiral ganglion neurons, interdental cells, and Claudius’ cells are lineally related to cells of the same type. We have also identified clonal relationships between organ of Corti cell types that reside on opposite sides of the tunnel of Corti. The extensive clonal dispersion detected and the observation of clonally related cells that span the tunnel suggest that progenitors integrating lineage label at ~E12 do not respect anatomical boundaries during convergent extension of the nascent cochlea. Correlating these observations with single cell transcriptomics of inner ear sensory cells may give us insight into the gene expression profiles required to specify individual sensory lineages and perhaps serve as the basis for new strategies to regenerate the sensory patch for therapeutic benefit.
Fetal pharmacotherapy for congenital deafness involves the use of gene replacement or drug strategies to restore wild type gene function during formation of the inner ear in utero. Our proof of concept for gene replacement focuses on the vesicular glutamate transporter 3 (VGLUT3) knockout mouse which is deaf at birth. Remarkably, adeno-associated virus (AAV)-mediated gene transfer of VGLUT3 into the E12.5 otocyst can restore auditory brainstem response thresholds (ABR) to near wild type levels. Our proof of concept for drug therapy focuses on the Usher 1c knockout mouse which is also deaf at birth. Transuterine microinjection of antisense oligonucleotide designed to correct mRNA slicing of harmonin incompletely but significantly restores ABR thresholds after a single dose to the E12.5 otocyst. These studies represent an exciting new experimental path for our lab that is refreshingly translational in perspective.
Our lab is a member of the Hearing Health Foundation’s Hearing Restoration Project (HRP). The organizing principle of the HRP is that strategic collaborations among participating labs will more efficiently advance the science needed to define efficacious therapies for treating hearing loss and tinnitus. Our specific role is to devise mouse model systems required to test candidate hair cell regeneration genes in the postnatal, deafened mouse inner ear. Our HRP affiliation has led to intellectually engaging and productive ongoing collaborations that leave us excited about future prospects for treating inner ear disease.
Selected Publications
- Brigande, J.V., Iten, L.E., and Fekete, D.M. (2000) A fate map of chick otic cup closure reveals lineage boundaries in the dorsal otocyst. Dev. Biol. 227: 256-270.
- Gubbels, S.P., Woessner, D.W., Mitchell, J.C., Ricci, A.J., and Brigande, J.V. (2008) Functional auditory hair cells produced in the mammalian cochlea by in utero gene transfer. Nature 455: 475-77. PMCID: PMC2925035
- Brigande, J. V. and Heller, S. (2009) Quo Vadis, Hair Cell Regeneration? Nature Neuroscience 12: 679-685. PMCID: PMC2875075
- Wang, L., Jiang, H., and Brigande, J.V. (2012) Gene transfer to the developing mouse inner ear by in vivo electroporation. Journal of Visualized Experiments 2012(64). PMID:22781586 p
- Jiang, H., Wang, L., Beier, K.T., Cepko, C.L., Fekete, D.M., and Brigande, J.V. (2013) Lineage analysis of the late otocyst stage mouse inner ear by transuterine microinjection of a retroviral vector encoding alkaline phosphatase and an oligonucleotide library. PloS One 8: e69314. PMID: 23935981
See PubMed for more of Dr. Brigande's publications.
Staff
Santhosh Chakkaramakkil Verghese Ph.D
Senior Research Associate
(503) 494-4568
Lorelei Millund
Research Assistant 2
(503) 494-4568
Stephen David, Ph.D.

Email: davids@ohsu.edu
Phone: 503-494-2931
Lab phone: 503-494-2969
Office: MRB 0934
Research Interests: auditory system, cortex, computational neuroscience, systems neuroscience, behavior
Stephen David joined the OHSU faculty in February 2012. Before coming to OHSU, he received his Ph.D. in Bioengineering from the University of California, Berkeley in 2006 and subsequently completed postdoctoral work in the Institute for Systems Research at the University of Maryland, College Park.
Summary of Current Research
Humans and other animals are exquisitely adept at creating a coherent sense of the world from complex and continuously changing sensory inputs. Throughout development, the brain's auditory system learns to categorize and discriminate important sounds, while ignoring irrelevant but often substantial noise. State of the art audio processing systems attempt to mimic these abilities, but even common sources of environmental noise severely confound automatic speech processors and distort the output of hearing aids and prosthetics. The David lab seeks to understand the neurophysiological and computational processes that underlie the remarkable abilities of the auditory brain, with an aim of understanding communication disorders and improving engineered systems for sensory signal processing.
Behavior-driven Changes in the Representation of Sensory Information
During normal behavior, important information can arrive from multiple sensory modalities, and the relevance of any given stimulus can change with behavioral demands. Thus the ability to robustly identify sensory events represents a combined effort of bottom-up multimodal representations and top-down control signals that extract sensory information appropriate to the task at hand. To understand these complementary processes, the lab conducts experiments that manipulate auditory attention and study how the cerebral cortex represents sounds under different behavior conditions. Data from these studies is used to develop computational models that integrate top-down and bottom-up processing under realistic, natural conditions.
Neural Representation of Natural auditory and Visual Stimuli
The David Lab is also interested in basic questions of how sensory information is represented by cortical neurons, especially under the rich and varied conditions encountered in the natural environment. Work from our lab has shown that the auditory cortex represents speech and other natural stimuli using algorithms that cannot be discerned from responses to the synthetic noise and tone stimuli typically used to characterize the auditory system. Ongoing studies aim to clarify how important natural signals are represented in cortex and to characterize the circuit mechanisms that produce these representations.
Selected Publications
- N. Ding, J.Z. Simon, S.A. Shamma, S.V. David. (2016) Encoding of natural sounds by variance of the cortical local field potential. J Neurophys, 115(5):2389-98.
- I.L. Thorson, J. Lienard, S.V. David. (2015) The essential complexity of auditory receptive fields. PLoS Comput Biol . 11(12):e1004628.
- S.J. Slee,. S.V. David. (2015) Rapid task-related plasticity of spectrotemporal receptive fields in the auditory midbrain. J Neurosci 35(38):13090-13102.
- M.J. McGinley, S.V. David, D.A. McCormick. (2015) Cortical membrane potential signature of optimal states for sensory signal detection. Neuron. 87(1):179-92.
- J.M. Stafford, B.R. Jarrett, O. Miranda-Dominguez, B.D. Mills, N. Cain, S. Mihalas, G.P. Lahvis, K.M. Lattal, S.H. Mitchell, S.V. David, J.D. Fryer, J.T. Nigg, D.A. Fair. (2014) Large-scale topology and the default mode network in the mouse connectome. Proc Natl Acad Sci U S A. 111(52):18745-50.
- N. Mesgarani, S.V. David, J.B. Fritz, S.A. Shamma. (2014) Mechanisms of noise robust representation of speech in primary auditory cortex. Proc Natl Acad Sci U S A. 111(18):6792-7
See more Dr. David's publications at PubMed.
David Lab
Lab Projects
Humans and other animals are exquisitely adept at creating a coherent sense of the world from the complex patterns that continuously bombard their senses. Throughout development, we identify patterns in the sounds around us and learn to categorize and discriminate important signals, while ignoring irrelevant but often substantial noise. State of the art audio processing systems attempt to mimic these abilities, but even the most common sources of environmental noise severely confound automatic speech processors and distort the output of hearing aids and prosthetics. We are interested in understanding the neurophysiological and computational processes that underlie the remarkable abilities of the auditory brain, with an aim of improving engineered systems for sensory signal processing.
Along a different intellectual line, but in a similar spirit of understanding complex systems, we also study the history of neuroscience and, in particular, how mentorship influences the transmission and evolution of ideas.
Behavior-driven changes in the representation of sensory informationConsider how the brain might represent an important natural sound, such as a vocalization, in a noisy environment. In more peripheral areas (left), such as primary auditory cortex, responses to both the vocalization and background noise are distributed broadly across the neural population (red indicates neurons that respond more to the vocalization and blue those that respond more to the noise). As information passes to subsequent stages, responses to the noise are suppressed and a smaller subset of neurons become more selective for the vocalization. Finally, in brain areas that execute decisions or motor responses (right), a small set of neurons encoding an appropriate behavior respond categorically only to the vocalization. The goal of understanding this process of hierarchical feature extraction lies at the core of our research.
During normal behavior, important information can arrive from multiple sensory modalities, and the relevance of any given stimulus can change with behavioral demands. Thus the ability to robustly identify sensory events represents a combined effort of bottom-up multimodal representations that are modulated by top-down down demands for information appropriate to the task at hand. To understand these processes, we conduct experiments that manipulate auditory attention and study how the cortex functions under these different behavior conditions. Data from these studies is used to develop computational models that integrate top-down and bottom-up processing under realistic, natural conditions.
Neural representation of natural auditory and visual stimuliWe are also interested in basic questions of how sensory information is represented by cortical neurons, especially under the rich and varied conditions encountered in the natural environment. Neural representations can be characterized by models, i.e., mathematical equations that describe the relationship between a sensory stimulus and subsequent neural activity. Much of our understanding of auditory brain representations is derived from experiments that probed neural response properties with simple synthetic stimuli, such as tones and noise bursts. While these experiments have revealed much about representation, particularly in more peripheral brain areas, recent experiments measuring neural responses to speech and other vocalizations have shown consistently that models based on responses to simple synthetic stimuli do not predict responses to the more complex natural stimuli. Instead, new and more comprehensive dynamical models are required to describe representations of these more behaviorally relevant stimuli.
With the continuous increase in available computational power, we have the ability to test and compare a huge variety of increasingly complex models. This new potential raises new issues: What is the best way to compare functional models of neurons? How should the large and diverse neurophysiological datasets be stored so that they can be available for testing new models? The Neural Prediction Challenge, a collaboration with Jack Gallant and Frederic Theunissen at UC Berkeley, is a database of single neuron recordings from auditory and visual systems using natural stimuli. Interested researchers can download the data and compare the performance of their model against other models fit with the same data. A related project, STRFpak, is a software package providing model estimation and validation tools that can be applied to any neurophysiological data set.
Effects of Hearing Loss on Central Auditory Representations
Hearing loss is not just a problem with the ear. Although most difficulty with hearing is caused by damage to the ear's hair cells or to the auditory nerve, the impact of reduced auditory input can cause lead to substantial compensatory plasticity in the brain areas responsible for processing sounds. This plasticity is particularly large if the hearing loss happens early in development. In such situations, when the brain does not wire normally, the benefits of restored hearing later in life (through hearing aids or cochlear implants) can be limited, as the brain is does not have the capability to process sounds normally. Recently, we have begun studying how natural sounds are represented in the auditory cortex after hearing loss and how hearing loss impacts the ability of top-down control systems to extract behaviorally relevant information from sounds.
History of Neuroscience
Neuroscience is a new but rapidly growing field, drawing on ideas and methodologies from many other research areas, including biology, psychology, physics, mathematics and philosophy. Depending on their training, each neuroscientist brings a unique perspective into their research. In ongoing research, we are studying how academic mentorship, the hands-on training received at the doctoral and postdoctoral level, influences the work of individuals and how training in multiple disciplines allows for the synthesis of different approaches into the new techniques that define neuroscience as its own field. Neurotree is a collaborative, open-access website that tracks and visualizes the academic genealogy. After nine years of growth driven by user-generated content, the site has captured information about the mentorship of over 45,000 neuroscientists. As a public resource, it has become a unique tool for a community of primary researchers, students, journal editors, and the press. The database captures a unique aspect of the history of the field, and it allows us to explore the evolution of new ideas and how mentorship has contributed their development. We are exploring new ways to improve the quality of the existing data and ways to link Neurotree to other datasets, such as publication and grant databases. Inspired by Neurotree's example, genealogies have been launched for a number of other fields under auspices of the Academic Family Tree, which aims to build a single genealogy across all academic fields.
See Dr. David's Laboratory of Brain, Hearing, and Behavior website.
Frederick (Erick) J. Gallun, Ph.D.

Email: gallunf@ohsu.edu
Phone: 503-494-4331
Lab phone: 503-494-5375
Office: HRC 0473
Research interests: psychoacoustics, auditory processing, binaural hearing, spatial hearing, temporal processing, traumatic brain injury, aging, hearing loss
Appointments
- Professor, Department of Otolaryngology-Head and Neck Surgery
- Faculty Member, Neuroscience Graduate Program
Background
Frederick J. Gallun, Ph.D. received his undergraduate degree from Reed College in 1993 and his Ph.D. from UC Berkeley in 2003, both in cognitive psychology. From 2003-2006, Gallun conducted an NIH-funded postdoctoral fellowship in Communication Disorders at Boston University. In 2006, he joined the VA RR&D National Center for Rehabilitative Auditory Research and turned his attention to the auditory processing problems of Veterans. In 2019, he joined the faculty of the Oregon Hearing Research Center and maintained a joint appointment with the NCRAR. He currently serves as the Chair of the American Speech-Language-Hearing Association Journals Board.
Summary of current research
Exposure to high-energy explosions (“blasts”), concussions and aging can all increase the risk of having trouble doing complicated listening tasks even when the ability to do simple auditory detection tasks is preserved. These findings have come out of studies conducted in Dr. Gallun’s lab that focus on the ability of listeners to perform complex listening tasks representative of the activities that comprise real world listening behavior.
Dr. Gallun and his colleagues are working to develop new ways to assess the ability to do complex auditory tasks and develop tools to improve or retrain those abilities. Tasks on which they are focused include the ability to listen to one of several people speaking at the same time. Participants range in age from 18-89 and have hearing that varies from very good to moderately poor. Some participants have been near explosions during their military career or have suffered blows to the head. Some of them have no complaints about their ability to hear and understand speech, while others find listening in noisy environments to be very difficult.

For more discussion of these issues, you are encouraged to listen to Dr. Gallun’s interview on CogNation, a podcast by Drs. Nelson and Hardy, two cognitive psychologists interested in the future of brain science and technology.
He recently presented a Keynote Speech at the Acoustical Society of America in Chicago, IL about his research career. You can view the recorded presentations here and the accompanying slides here.
Selected publications
- Gallun, F.J., Srinivasan, N. K., Diedesch, A. C. (2021). “Clinical Ramifications of Hearing Impairment and Aging on Spatial and Binaural Hearing” in Binaural Hearing, edited by Litovsky, R.Y., Goupell, M.J., A.N. Popper and R.R. Fay. Springer Handbook of Auditory Research, Volume 73. ASA Press and Springer Nature Switzerland, Cham, Switzerland
- Larrea-Mancera, E. S., Stavropoulos, T., Hoover, E. C., Eddins, D. A., Gallun, F.J., & Seitz, A. R. (2020). Portable Automated Rapid Testing (PART) for auditory assessment: Validation in a young adult normal-hearing population. Journal of the Acoustical Society of America. 148(4) 1831-1851 https://doi.org/10.1121/10.0002108
- Gallun, F.J., Best, V. (2020). “Age-Related Changes in Segregation of Sound Sources” in Aging and Hearing: Causes and Consequences, edited by K.S. Helfer, E.L. Bartlett, A.N. Popper and R.R. Fay. Springer Handbook of Auditory Research, Volume 72. ASA Press and Springer Nature Switzerland, Cham, Switzerland
- Gallun, F.J., Jakien, K.M. (2019) “The ability to allocate attentional resources to a memory task predicts speech-on-speech masking for older listeners” Proceedings of the International Congress on Acoustics, Germany, September 2019 http://pub.dega-akustik.de/ICA2019/data/articles/000007.pdf
- Hoover, E.C., Kinney, B.N, Bell, K.L., Gallun, F.J., Eddins, D.A. (2019) "A comparison of behavioral methods for indexing the auditory processing of temporal fine structure cues" Journal of Speech, Language, and Hearing Research, 62(6), 2018-2034 https://pubs.asha.org/doi/full/10.1044/2019_JSLHR-H-18-0217
- Papesh, M. A., Elliott, J. E., Callahan, M. L., Storzbach, D., Lim, M. M., & Gallun, F. J. (2019). Blast exposure impairs sensory gating: Evidence from measures of acoustic startle and auditory event-related potentials. Journal of neurotrauma. Online ahead of print. https://doi.org/10.1089/neu.2018.5801
- Jakien, K.M. and Gallun, F.J. (2018) “Normative data for a rapid, automated test of spatial release from masking” American Journal of Audiology, 27, 529-538. https://doi.org/10.1044/2018_AJA-17-0069
- Souza, P.E., Wright, R., Gallun, F.J. (2018) “Reliability and repeatability of the speech cue profile” Journal of Speech, Language and Hearing Research, 61(8):2126-2137. https://doi.org/10.1044/2018_JSLHR-H-17-0341
- Souza, P.E., Hoover, E.C., Blackburn, M., Gallun, F.J. (2018) “The characteristics of adults with severe hearing loss” Journal of the American Academy of Audiology, 29(8):764-779. https://doi.org/10.3766/jaaa.17050
- Gallun, F.J., Lewis, M.S., Papesh, M.A. (2017) “Hearing complaints among veterans following traumatic brain injury” Brain Injury. 31(9), 1183-1187. http://dx.doi.org/10.1080/02699052.2016.1274781
- Adler, H.J., Anbuhl, K.L., Atcherson, S.R., Barlow, N., Brennan, M.A., Brigande, J.V., Buran, B.N., Fraenzer, J.T., Gale, J.E., Gallun, F.J. and Gluck, S.D. (2017) “Community network for deaf scientists” Science, 356 (6336), 386-387. https://doi.org/10.1126/science.aan2330
- Hoover, E., Souza, P.E., Gallun, F.J. (2017) “Auditory and cognitive factors associated with speech-in noise complaints following mild traumatic brain injury” Journal of the American Academy of Audiology. 28(4): 325-339. https://doi.org/10.3766/jaaa.16051
- Papesh, M.A., Folmer, R.L., Gallun, F.J., (2017) “Cortical measures of binaural processing predict spatial release from masking performance” Frontiers in Human Neuroscience, 11:124 http://dx.doi.org/10.3389/fnhum.2017.00124
- Ellinger, R.L., Jakien, K.M., Gallun, F.J. (2017) “The role of interaural differences in speech intelligibility in complex multitalker environments” Journal of the Acoustical Society of America, 141 EL170 http://doi.org/10.1121/1.4976113
- Gallun, F.J., Lewis, M.S., Folmer, R.L., Hutter, M., Papesh, M.A., Belding, H., Leek, M.R. (2016) “Chronic effects of exposure to high-intensity blasts: Results on tests of Central Auditory Processing” Journal of Rehabilitation Research and Development, 53(6): 705-720.
- Srinivasan, N.K., Jakien, K.M, Gallun, F.J. (2016) “Release from masking for small separations: Effects of age and hearing loss” Journal of the Acoustical Society of America, 140, EL73. http://doi.org/10.1121/1.4954386
- Souza, P.E., Wright, R., Gallun, F.J. (in press) “Contributions to speech-cue weighting in older adults with impaired hearing” Journal of Speech, Language, and Hearing Research
Gallun Lab
Lab Phone: 503-494-5375
Mailing address: Erick Gallun Lab
Oregon Health & Science University
Oregon Hearing Research Center
Mail Code: NRC04
3181 SW Sam Jackson Park Road
Portland, OR 97239
Current Lab Members
Lauren Charney
Research Audiologist
Nicole Dean
Research Assistant
Conner Corbett
Research Assistant
Karen Garica
Student Worker
Current Projects
EEPET
Enhanced Environments for Psychophysical Evaluation and Training (EEPET) aims to address the difficulty many people have hearing speech in noise by improving testing to include more realistic environments which increase motivation and correlate more strongly with self-report. Current test methods do not accurately reflect how people report performing in real-world situations, and by improving these test methods, we hope to improve training and rehabilitation methods.
Find the StudyPage for this project here.
MAMAT
Mediators and Moderators of Auditory Training (MAMAT) aims to develop an auditory training program which could mitigate the hearing issues that are currently thought to influence the development of Alzheimer’s and related dementias.
Check this page later for a link to our StudyPage.
Angie Garinis, Ph.D.

Email: garinis@ohsu.edu
Phone: 503-494-5019
Office: HRC 0472
Research Interests: ototoxicity; auditory development; physiologic measures of hearing
Background: Dr. Angela Garinis joined the Department of Otolaryngology and the Oregon Hearing Research Center in 2011. Dr. Garinis received her Master’s degree in clinical audiology and a Ph.D. in Speech & Hearing Sciences at the University of Arizona in Tucson. She completed a three-year NIH funded postdoctoral fellowship at the University of Washington in Dr. Lynne Werner’s Infant Psychoacoustics Laboratory (2008-2011). Her primary research interests involve improving non-invasive techniques for the diagnoses of middle-ear and/or cochlear dysfunction. She also has a strong interest in the diagnoses and monitoring of ototoxic hearing loss in patients with cystic fibrosis receiving aminoglycoside treatments. She currently has NIH-NIDCD funding to investigate the effects of intravenous aminoglycoside treatments on the cochlear and medial efferent auditory system in patients with cystic fibrosis.
NCRAR website has selected publications: https://www.ncrar.research.va.gov/AboutUs/Staff/Garinis.asp
Lina A.J. Reiss, Ph.D.

Email: reiss@ohsu.edu
Phone: 503-494-2917
Lab phone(s): 503-494-5868
503-494-2996
Office: HRC 0474
Research Interests: cochlear implants, hearing aids, brain plasticity, hearing loss, auditory perception, psychoacoustics, auditory neuroscience
Background
Lina Reiss received her Ph.D. in Biomedical Engineering from the Johns Hopkins University in 2005, with a focus on neurophysiology of sound localization circuits in the auditory brainstem. She holds a B.S.E. in Mechanical Engineering from Princeton University. After receiving her Ph.D., she spent five years as a postdoctoral fellow in the area of cochlear implants and psychoacoustics at the University of Iowa. In 2010, she was appointed as faculty in the Department of Otolaryngology and the Oregon Hearing Research Center, with joint appointments in Departments of Biomedical Engineering and Behavioral Neuroscience.
Summary of Current Research
Cochlear implants are a surgically implantable device that bypasses damaged auditory cells and directly stimulates the auditory nerve as a treatment for those with severe-profound hearing loss. Cochlear implants have been successful in restoring sound awareness and speech perception for many patients, but there is significant variability in outcomes, and many cochlear implantees have difficulty with speech in background noise. Lina Reiss and colleagues study ways to improve these outcomes through a variety of approaches.
There are two laboratories: a human clinical research laboratory, and neurophysiology/cochlear implant design laboratory. In the clinical lab, we study pitch and speech perception in human subjects with cochlear implants and/or hearing aids, with an emphasis on 1) combined acoustic and electric stimulation and 2) plasticity and how plasticity can shape perceptual responses and be used to improve outcomes. In the neurophysiology lab, we are currently studying whether electrical stimulation causes hearing loss, and if so, what designs or treatments may reduce hearing loss. A future project is to measure plasticity of tonotopic representations in response to long-term cochlear implant stimulation in the auditory brainstem.
For more information, see the Reiss Cochlear Implant and Hearing Aid Research Lab.
Selected Pubications
- Zhang, H., Stark, G., and Reiss, L.A. (2015) Changes in gene expression and hearing thresholds after cochlear implantation. Otol. Neurotol., 36(7):1157-65.
- Reiss, L.A.J., Ito, R.A., Eggleston, J.L., Liao, S., Becker, J.J., Lakin, C.E., Warren, F.M., and McMenomey, S.O. (2014) Pitch adaptation patterns in bimodal cochlear implant users: Over-time and after experience. Ear. Hear. [Epub ahead of print]
- Tanaka, C., Nguyen-Huynh, A., Loera, K., Stark, G., and Reiss, L. (2014) Factors associated with hearing loss in a normal-hearing guinea pig model of hybrid cochlear implants. Hear. Res. 316: 82-93.
- Reiss, L.A.J., Ito, R.A., Eggleston, J.L., and Wozny, D.R. (2014) Abnormal binaural spectral integration in cochlear implant users. J. Assoc. Res. Otolaryngol., 15(2):235-48.
- Reiss, L.A., Turner, C.W., Karsten, S.A., and Gantz, B.J. (2013) Plasticity in human pitch perception induced by tonotopically mismatched electro-acoustic stimulation. Neuroscience, 256: 43-52.
- Reiss, L.A., Lowder, M.L., Karsten, S.A., Turner, C.W., and Gantz, B.J. (2011) "Effects of extreme tonotopic mismatches between bilateral cochlear implants on electric pitch perception: A case study". Ear Hear. 32(4):536-40.
- Gantz, B.J., Hansen, M.R., Turner, C.W., Oleson, J.J., Reiss, L.A., and Parkinson, A.J. (2009) "Hybrid 10 clinical trial: preliminary results". Audiol. Neurootol. 14 Suppl 1:32-38.
- Turner, C.W., Reiss, L.R. and Gantz, B. (2008). "Combined acoustic and electric hearing: Preserving residual acoustic hearing." Hear. Res. 242(1-2): 164-171.
- Reiss, L.A.J., Bandyopadhyay, S., and Young, E.D. (2007) "Effects of stimulus spectral contrast on receptive fields of dorsal cochlear nucleus neurons". J. Neurophys. 98(4):2133-2143.10.
- Reiss, L.A.J., Turner, C.W., Erenberg, S.R., and Gantz, B.J. (2007), "Changes in pitch with a cochlear implant over time", J. Assoc. Res. Otolaryngol. 8(2): 241-257.
- 11. Reiss, L.A.J., and Young, E.D. (2005), "Spectral edge sensitivity in neural circuits of the dorsal cochlear nucleus", J. Neurosci. 25(14): 3680-3691.
- 12. Graziano, M.S.A., Reiss, L.A.J., and Gross, C.G. (1998), "A neuronal representation of the location of nearby sounds", Nature 397, 428-430.
See Dr. Reiss's publications at PubMed
Reiss Lab
Please access Dr. Reiss's lab page here.
Tianying Ren, M.D.

Email: rent@ohsu.edu
Phone: 503-494-2938
Office: HRC 0416
Lab Phone: 503-494-2934
Research Interests: cochlear mechanics, otoacoustic emissions, auditory physiology
Background
Tianying Ren completed his medical and graduate education (M.D., M.S.) at Xi'an Medical University (now School of Medicine of Xi'an Jiaotong University) in 1983. Following his residency, he worked as a lecturer in the Department of Otolaryngology at the First Teaching Hospital of Xi'an Medical University. He was a World Health Organization (WHO) fellow and a research investigator in Kresge Hearing Research Institute at the University of Michigan (1990-1995). He joined Oregon Hearing Research Center at Oregon Health & Science University in 1996.
Summary of current research
The Ren lab currently studies the mechanism of the cochlear amplification by measuring cochlear micromechanical responses to acoustical and electrical stimulations in normal and genetically modified cochleae using custom-built low-coherence heterodyne interferometers. The new data from these measurements will provide crucial information for understanding how the cochlea achieves its extraordinary sensitivity, frequency selectivity, and dynamic range.
The other research area of the Ren lab is on the generation and propagation of otoacoustic emissions. The objective of this study is to determine the cellular origin and sub-cellular mechanisms responsible for generating distortion product otoacoustic emissions (DPOAEs) by conducting a series of novel in vivo experiments using a scanning low-coherence interferometer. This project will provide new knowledge for improving applications of DPOAEs as a non-invasive tool for diagnosing hearing loss in humans and for studying cochlear mechanics in experimental animals.
Selected Publications
- He, W., Kemp, D., Ren, T. Timing of the reticular lamina and basilar membrane vibration in living gerbil cochleae. eLife Sci. 7, 37625 (2018).
- Ren, T., He, W. & Kemp, D. Reticular lamina and basilar membrane vibrations in living mouse cochleae. Proc. Natl. Acad. Sci. USA. 113, 9910-9915 (2016).
- Ren, T., He, W. & Barr-Gillespie, P. G. Reverse transduction measured in the living cochlea by low-coherence heterodyne interferometry. Nat Commun 7, 10282 (2016).
- Ren, T., He, W. & Gillespie, P. G. Measurement of cochlear power gain in the sensitive gerbil ear. Nat. Commun. 2, 216 (2011).
- He, W., Fridberger, A., Porsov, E., Grosh, K. & Ren, T. Reverse wave propagation in the cochlea. Proc. Natl. Acad. Sci. USA. 105, 2729-2733 (2008).
- Ren, T. Reverse propagation of sound in the gerbil cochlea. Nat Neurosci 7, 333-334 (2004).
- Ren, T. Longitudinal pattern of basilar membrane vibration in the sensitive cochlea. Proc. Natl. Acad. Sci. USA. 99, 17101-17106 (2002).
Please see Dr. Ren's publications at PubMed.
Xiaorui Shi, M.D., Ph.D.

Email: shix@ohsu.edu
Phone: 503-494-2997
Lab phone: 503-494-7149
Office: HRC 0426
Research Interests: cochlear homeostasis, blood-labrynth barrier, stria vascularis, pericytes
Background
Xiaorui Shi, M.D., Ph.D., is a Professor with the Oregon Hearing Research Center. Dr. Shi joined OHSU in 2005.
Overview
Appointments:
Current appointments:
Professor in the Department of Otolaryngology-Head and Neck Surgery
Degrees:
- M.D. degree from Henan Medical University, Henan, China:
- M.S. degree from Shanghai Second Medical University, Shanghai, China
- Ph.D. degree from Henan Medical University, Henan, China
Research Interests
Normal hearing requires the blood supply to the cochlea is well-regulated and the blood-labyrinth barrier tightly-controlled. The structural integrity of the microvasculature is critical to its function in maintaining the supply of oxygen, nutrients, and hormones to our ears, and for removing toxins and metabolic waste products. Insufficient blood supply to the ear is among the primary causes of age-related and noise-induced hearing loss, and is implicated in autoimmune inner ear disease and several genetically-linked hearing diseases as well. The aim of my lab is to obtain a better understanding of the mechanisms by which blood flow to the ear is regulated, blood-labyrinth barrier is controlled, and compromised vasculature is restored after damage. Ultimately, our goal is to develop new treatments, ameliorate damage, and improve the quality of life for people who suffer from vascular dysfunction related hearing loss.
- Regulation of cochlear blood flow
- Cellular mechanisms controlling the integrity of the blood-labyrinth-barrier
- Vascular pathology in aging, loud sound, and inflammatory induced damage to the ear
- Vascular remodeling and angiogenesis after damage
Significant Discoveries and Advances
- A fibrocyte-pericyte coupling controls regional cochlear blood flow
- Perivascular resident macrophages in the cochlear blood-labyrinth barrier renew via migration of bone-marrow-derived cells
- Pericyte plasticity in response to loud-sound microvessel damage
- Bone-marrow cells are recruited to the acoustically damaged blood-labyrinth barrier
- Mass spectroscopy shows a high percentage of proteins in purified mouse strial capillaries are transporters
- Developed a novel "sandwich-dissociation" method for isolation of strial capillaries and a "mini-chip" method for isolation and primary culture of strial blood-labyrinth-barrier component cells″ Breakdown of the strial BLB, a common feature in a wide range of pathological conditions (cochlear inflammation, loud sound trauma, and ageing), can be remodeled by stem cells
News
Dr. Shi was the recipient of the 2016 OHSU Technology Transfer & Business Awards, for having your technology, OHSU #2188, Cochlear endothelial cells (ECs), pericytes (PCs) and perivascular resident macrophage-like melanocytes (PVM/Ms) cell line, licensed in fiscal year 2016.
Selected publications
- Shi, X. (2016). Pathophysiology of the cochlear intrastrial fluid-blood barrier (review). Hear Res, doi:10.1016/j.heares.2016.01.010.
- Zhang, J., Chen, S., Hou, Z., Cai, J., Dong, M., & Shi, X. (2015). Lipopolysaccharide-induced middle ear inflammation disrupts the cochlear intra-strial fluid–blood barrier through down-regulation of tight junction proteins. PLoS One, 10(3), e0122572.
- Neng, L., Zhang, J., Yang, J., Zhang, F., Lopez, I. A., Dong, M. & Shi, X. (2015). Structural changes in thestrial blood–labyrinth barrier of aged C57BL/6 mice. Cell Tissue Res, 361(3), 685-696.
- Wilson, T., Omelchenko, I., Foster, S., Zhang, Y., Shi, X. & Nuttall, A. L. (2014). JAK2/STAT3 inhibition attenuates noise-induced hearing loss. PLoS One, 9(10), e108276.
- Shi, X., Zhang, F., Urdang, Z., Dai, M., Neng, L., Zhang, J., Chen, S., Ramamoorthy, S. & Nuttall, A. L. (2014). Thin and open vessel windows for intra-vital fluorescence imaging of murine cochlear blood flow. Hear Res, 313, 38-46.
- Zhang, F., Dai, M., Neng, L., Zhang, J. H., Zhi, Z., Fridberger, A. & Shi, X. (2013). Perivascular macrophage-like melanocyte responsiveness to acoustic trauma—a salient feature of strial barrier associated hearing loss. FASEB J, 27(9), 3730-3740.
- Zhang, F., Zhang, J., Neng, L., & Shi, X. (2013). Characterization and inflammatory response of perivascular-resident macrophage-like melanocytes in the vestibular system. J Assoc Res Otolaryngol, 14(5), 635-643.
- Neng, L., Zhang, W., Hassan, A., Zemla, M., Kachelmeier, A., Fridberger, A., Auer, M. & Shi, X. (2013). Isolation and culture of endothelial cells, pericytes and perivascular resident macrophage-like melanocytes from the young mouse ear. Nat Prot, 8(4), 709-720.
- Zhang, W., Dai, M., Fridberger, A., Hassan, A., DeGagne, J., Neng, L., Zhang, F., He, W., Ren, T., Trune, D. Auer, M. & Shi, X.R. (2012). Perivascular-resident macrophage-like melanocytes in the inner ear are essential for the integrity of the intrastrial fluid–blood barrier. Proc Nat'l Acad Sci U S A, 109(26), 10388-10393.
- Dai, M, Yang, Y., Omelchenko, I., Nuttall, A.L., Kachelmeier, A., Xiu, R. & Shi, X.R. (2010). Bone marrow cell recruitment for repair of acoustically damaged blood-labyrinth-barrier mediated by a local iNOS/SDF-1α signal pathway. Am J Path, 177(6):3089-99. PMC2993278. Am J Pathol. 2009 May;174(5):1692-704.
- Shi X. (2009) Cochlear pericyte responses to acoustic trauma and the involvement of hypoxia-inducible factor-1alpha and vascular endothelial growth factor. Am J Path, 174(5):1692-1704.
Please see Dr. Shi's full list of publications at PubMed.
Shi Lab
Dr. Shi's Inner Ear Blood Flow Lab
Normal cochlear blood flow is critical for generating the endocochlear potential (EP) on which transduction in hair cells depends. Disruption of cochlear blood flow occurs in a wide variety of hearing disorders including loud sound-induced hearing loss (endothelial injury), ageing-related hearing loss (lost vascular density), and genetic hearing loss (Norrie disease: strial avascularization). Progression of blood flow pathology often parallels progression in hair cell loss and hearing impairment. To sustain hearing acuity, a healthy blood flow must be maintained. The blood supply not only provides energy to the organ, but is also a source of hormones and neurotrophic factors for maintaining organ health. A better understanding of the underlying mechanisms will facilitate the development of effective drug therapies for vascular dysfunction-related hearing loss. Hearing loss, accompanying with tinnitus and sound hypersensitivity, is a common condition which leads to communication problems and social isolation. The ultimate goal of Dr. Shi's research is to improve the quality of life of people with vascular dysfunction associated metabolic deafness. Under NIH grant support and In active collaboration with a number of groups both within OHRC and outside OHSU, Dr. Shi's lab has established a broad range of advanced techniques, including intra-vital microscopy for blood flow imaging in a living animal, systemic transplantation of bone marrow cells, a 'sandwich-pressure' method for isolating strial capillaries, a 'mini-chip' method for isolating and culturing BLB component cells, co-culture in 3D matrix gel, RNA-seq (RNA Sequencing), mass spectrometry, and siRNA transfection, and measurement of vascular permeability with multiple tracers, and advanced transmission electron microscopy. With these established research approaches, Dr. Shi's have progressively begun to gain a better understanding of how cochlear blood flow and the blood-labyrinth-barrier are regulated and discovered unique feature of stiral vascular system.

Research Projects in Dr. Shi's Lab
Inner Ear Blood Flow Regulation
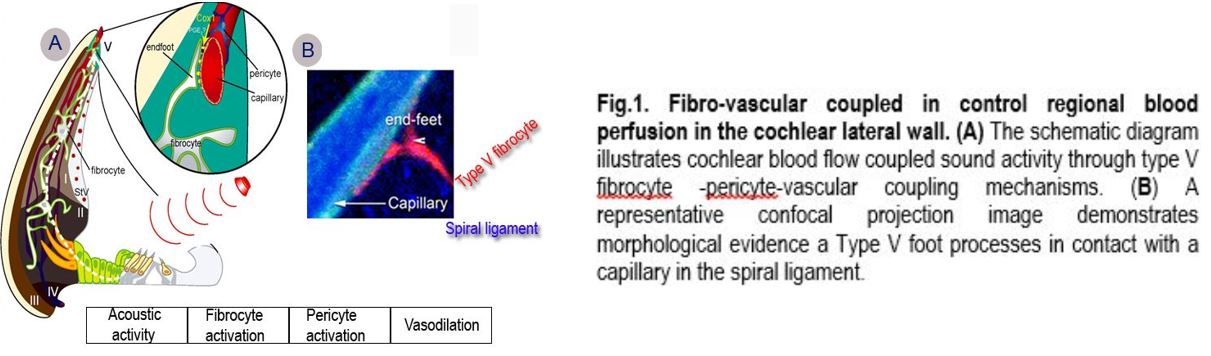
The cochlea is a high energy demand organ which transduces acoustic input to electrical signals within a time scale of microseconds. The transduction critically depends on adequate vascular support to provide a sufficient supply of oxygen. Regulation of cochlear blood flow includes central neural and local and auto-regulatory pathways at the level of artery, arterioles and capillaries. Dr. Shi's lab particular focuses on myogenic properties of pericytes in the spiral ligaments in controlling capillary blood flow and maintaining microvascular homeostasis. The pericytes in the spiral ligament express contractile proteins, including α-smooth muscle actin and tropomyosin, and exhibit vasocontractility under both in vivo and in vitro conditions. The contractility of pericytes affects flow resistance of the vascular network, and alters overall blood flow. Pericytes may be also functionally linked to form a "pumping system" to regulate blood flow. A recent experiment from Dr. Shi's lab has demonstrated that cochlear blood flow is modulated by lateral fibrocyte input (Fig.1). Fibrocytes have long been regarded to facilitate generation of the endocochlear potential by recycling K+ from hair cell transduction, through gap junctions to strial intermediate cells and marginal cells, into the endolymph. Dr. Shi's lab discovered a novel that cochlear blood flow (CBF) is regulated by a fibro-vascular (type V fibrocyte-pericyte) coupled control mechanism signaled by both lactate, acting through monocarboxylate transporter 1, and nitric oxide, produced by neuronal nitric oxide synthase. The mechanisms underlying the pathophysiology of cochlear blood flow is of fundamental clinical importance. A better understanding of cochlear blood flow (CoBF) will enable more effective management of hearing disorders resulting from aberrant blood flow. Dr. Shi's laboratory is actively in the study of regional signals in control of cochlear blood flow and change of blood flow under various pathological conditions such as loud sound stimulation.
Blood-Labyrinth-Barrier Pathophysiology
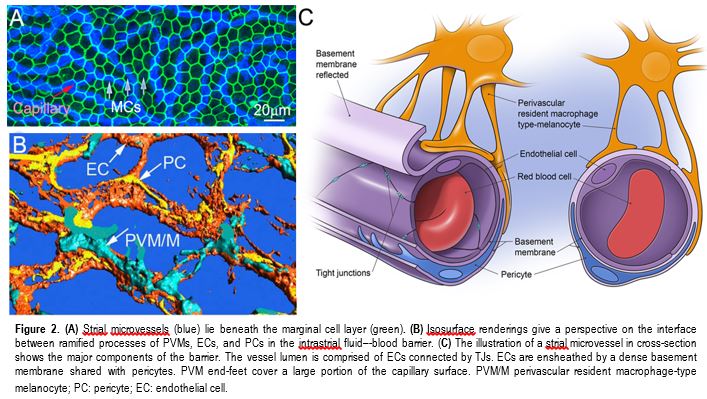
In the classic view, the BLB in the stria vascularis (strial BLB) is comprised of endothelial cells and an underlying basement membrane. Endothelial cells connect to each other by tight junctions and form a diffusion barrier which selectively excludes most blood-borne substances from entering the ear, protecting it from systemic influences. The Shi lab has shown the strial BLB is more complex. In addition to the microvascular endothelium, basement membrane, and pericytes, the strial BLB includes a substantial number of perivascular resident macrophages (PVMs). All together these cell types constitute a unique "cochlear vascular unit" (Fig.2). Signal communication between the cells may be critical for restricted permeability and transport, and for providing a proper environment for hearing function. Signaling between PCs and PVMs is essential for controlling the permeability of the BLB (Fig.3). The lab uses both in vivo and in vitro cell line based models to actively study cellular mechanisms controlling the permeability of strial BLB and changes in the BLB with aging, loud sound exposure, and inflammation
Vascular Remodeling and Angiogenesis
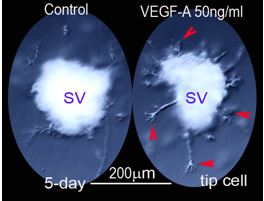
Can damaged or degenerated vessels be regenerated in the ear? The Shi lab recently discovered a rich population of neural glial antigen 2 (NG2+)/vimentin+/nestin+ perivascular cells (mesenchymal stem cells) in the strial capillary network. Do these cells have angiogenic potential and serve as vascular progenitors? The question is timely since vascular injury, vascular density loss, and avascularization are seen in a wide variety of hearing disorders, including in loud sound-induced hearing loss, ageing-related hearing loss (lost vascular density), and genetic hearing loss (Norrie disease). Is restoration of cochlear blood flow critical for stabilizing or slowing hearing decline (e.g., presbycusis) and facilitating restoration of hearing function (e.g., loud sound-induced hearing loss)? To answer these questions, the Shi lab has created an in vivo pericyte depletion/vascular degeneration mouse model. With this model, in association with a newly established ex vivo tissue based-3D matrigel matrix model, the lab will investigate the relationship between vascular function, cell survival, endocochlear potential, and hearing. The lab will also determine the molecular signaling which initiates the angiogenesis in the stria vascularis needed for restoration of cochlear vascular function.
Past & Present International Visiting Scholars, PhD Students, and Postdoctoral Students in the Shi lab
Yunpei Zhang
Senior Research Associate
Pingting Wang
Research Assistant 2

Dennis Trune, Ph.D., Professor

Email: truned@ohsu.edu
Phone: 503-494-8032
Office: HRC 0416
Research Interests: immune-mediated inner-ear disease and hearing loss, intratympanic delivery of steroids for improved recovery of hearing loss, development of novel treatments for hearing loss, ion and water transport dysfunction in different forms of hearing loss, control of inflammatory processes of the middle ear, impact on middle-ear inflammation on the inner ear
Dennis R. Trune, Ph.D., M.B.A., is a Professor in the Oregon Hearing Research Center. He joined OHSU in 1983.
Overview
Current Appointments
- Professor in the Department of Otolaryngology-Head and Neck Surgery
Previous Appointments
- Professor in the Department of Otolaryngology-Head and Neck Surgery
- Adjunct Researcher with the National Center for Rehabilitative Auditory Research, Portland VAMC
Degrees
- B.A. degree in biology from the University of Michigan-Flint
- M.S. degree in biology from Northern Arizona University
- M.B.A. degree from Portland State University
- Ph.D. degree in anatomy from Louisiana State University Medical School; conducted dissertation research at the Kresge Hearing Research Laboratory
- Postdoctoral fellowship with Dr. David Lim in the Otolaryngology Department at Ohio State University
Summary of Current Research
Research Interests
- Immune-mediated inner-ear disease and hearing loss
- Intratympanic delivery of steroids for improved recovery of hearing loss
- Development of novel treatments for hearing loss
- Ion and water transport dysfunction in different forms of hearing loss
- Control of inflammatory processes of the middle ear
- Impact on middle-ear inflammation on the inner ear
Major Milestones and Significant Discoveries
- New treatment for sudden and immune-mediated hearing loss
- Demonstration that inflammatory processes occur in the inner ear with middle-ear infections
- Identification of fluid-clearing mechanisms in the middle-ear compromised during infections, leading to the painful effusions found in otitis media
- Identification of potential gene defects that cause middle-ear infections
Laurence Trussell, Ph.D.

Email: trussell@ohsu.edu
Phone: 503-494-3424
Lab phone: 503-494-3409
Office: MRB 0924
Research Interests: synaptic physiology, auditory, hearing, receptors, synapses, neurotransmitters, plasticity
Background
Laurence Trussell received his Ph.D. in Biology from the University of California, Los Angeles in 1983. After initial postdoctoral work at UCLA, he obtained further training at Washington University, St. Louis. In 1990, he received a faculty appointment at the University of Wisconsin, Madison. In 1999, he was appointed as professor in the Oregon Hearing Research Center with an appointment as scientist at the Vollum Institute.
Overview
Current appointments:
- Professor in the Department of Otolaryngology-Head and Neck Surgery
- Scientist with the Vollum Institute
Previous appointment:
- Associate Professor in the Department of Physiology, University of Wisconsin, Madison
Degrees:
- B.A. degree in biology from University of California-San Diego
- Ph.D. degree in biology from University of California-Los Angeles
- Postdoctoral work at UCLA and Washington University
Summary of current research
Research Interests
- Brainstem circuits that process acoustic signals
- Synaptic physiology - how do cells and synapses preserve information provided by the ear?
Major Milestones and Significant Discoveries
- Neurons that encode timing of acoustic signals are molecularly and biophysically specialized for that function
- Cellular/synaptic learning mechanisms occur even at the earliest levels of sensory processing
- Corelease of multiple transmitters from neurons act together on the same receptor
- Ca2+ channels are expressed in axons and control the triggering of complex spike activity.
In addition to his appointment with the Oregon Hearing Research Center, Dr. Trussell is a Senior Scientist with the Vollum Institute. To access his publications,
Click here to go to PubMed.
Selected Publications
- Apostolides, P. F., & Trussell, L. O. (2013). Regulation of interneuron excitability by gap junction coupling with principal cells. Nat Neurosci, 16(12), 1764-1772.
- Apostolides, P. F., & Trussell, L. O. (2014). Control of interneuron firing by subthreshold synaptic potentials in principal cells of the dorsal cochlear nucleus. Neuron, 83(2), 324-330.
- Huang, H., & Trussell, L. O. (2014). Presynaptic HCN channels regulate vesicular glutamate transport. Neuron, 84(2), 340-346.
- Borges-Merjane, C., & Trussell, L. O. (2015). ON and OFF unipolar brush cells transform multisensory inputs to the auditory system. Neuron, 85(5), 1029-1042.
- Lu, H. W., & Trussell, L. O. (2016). Spontaneous Activity Defines Effective Convergence Ratios in an Inhibitory Circuit. J Neurosci, 36(11), 3268-3280.
- Tang, ZQ and Trussell, LO (2017) Serotonergic modulation of sensory representation in a central multisensory circuit is pathway-specific. Cell Reports, 20, 1844–1854
- Lu, HW, Balmer, TS, Romero, GE, and Trussell LO (2017) Slow AMPAR synaptic transmission is determined by stargazin and glutamate transporters. Neuron, 96, 73-80.
- Irie, T and Trussell, LO (2017) Double nanodomain coupling of calcium channels, ryanodine receptors and BK channels controls generation of burst firing. Neuron, 96, 856-870.
- Moore, AK, Weible, AP, Balmer, TS, Trussell, LO, Wehr, M. (2018) Rapid rebalancing of excitation and inhibition in cortical circuits. Neuron, 97:1341-1355.
- Joris, PX and Trussell LO (2018) The calyx of Held: a hypothesis on the need for reliable timing in an intensity difference encoder. Neuron, 100:534-549.
- Balmer, TS and Trussell LO (2019) Selective Targeting of unipolar brush cell subtypes by cerebellar mossy fibers. Elife 8.pii:e44964.
Trussell Lab
Please access Dr. Trussell's Lab page here.