Schnell Lab
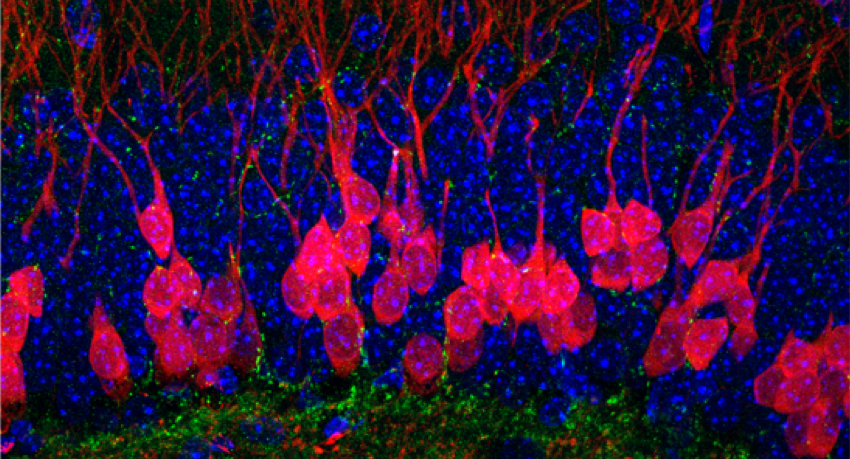
Research interests
Our current research focuses on how synapses are formed in the brain by adult born neurons, how these neurons contribute to neuronal circuit function, and how they contribute to recovery or the development of neurologic disease after neurologic injury. Newly generated neurons must be integrated into existing neuronal circuits to contribute to information processing, and alterations in this process could have profound clinical implications. Our current focus is on understanding how adult-born neurons might contribute to the pathogenesis of post-traumatic epilepsy, and we have developed techniques to analyze synaptic transmission at synapses formed by these adult-born cells and to evaluate the behavioral consequences of activity in these adult-born neurons. Our scientific expertise lies in the study of neuronal structure and synaptic function, using electrophysiology, molecular biology, translational models and immunohistochemical/anatomical imaging techniques.
Ongoing projects
-
By combining optogenetics and transgenic labeling techniques, several projects in the lab examine hippocampal circuit plasticity and rewiring during the development of epilepsy. In one recent study, we found that adult-born neurons contribute to the formation of recurrent circuits, which might contribute to hippocampal hyperexcitability in epilepsy. Our hope is to not only describe how specific neuronal circuit elements are altered during epileptogenesis, but also to determine whether these changes contribute to, or help ameliorate, hippocampal hyperexcitability.
-
Although the brain has some capacity for regeneration after injury through the enhancement of adult neurogenesis, we have discovered that neurons born into an injured brain develop aberrantly and may contribute to circuit dysfunction. Ongoing studies suggest that anesthetic drugs might modulate this aberrant neurogenesis, and might have therapeutic benefits if administered early after injury. We are combining imaging, electrophysiology, and behavioral assays with translational models of brain injury to evaluate both mechanisms and potential treatments for post-traumatic neural circuit dysfunction.
-
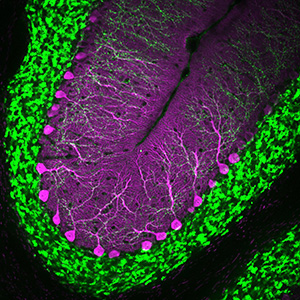
The alpha-2 delta protein family plays a role in neuronal Ca++ channel function, synaptic transmission, and synaptogenesis. We are interested in how these various roles interact during neuronal development and, and are engaged in both in vivo and in vitro studies of the fascinating protein family
Our people
-

Eric began his research career in Michael Hasselmo’s laboratory at Harvard, studying the cholinergic modulation of synaptic transmission. He then obtained his Ph.D. in Roger Nicoll’s lab at UCSF, and helped establish the role of PSD95 in synapse formation and AMPA receptor trafficking to synapses. He arrived at the Portland VA/OHSU in 2008, and began his faculty career with mentorship from Gary Westbrook at the Vollum Institute, studying the mechanisms underlying the maturation and synaptic integration of adult-born granule cells. He has since established his own lab, but maintains a close collaboration with Gary on a number of ongoing projects. Outside of the lab, Eric is an avid bike commuter and loves spending time on the water, either liquid or frozen.
-

Natasha is an MD/PhD student who joined the Schnell lab in 2020. She grew up in Cincinnati, Ohio and went to college at Washington University in Saint Louis, where she pursued a degree in philosophy, neuroscience and psychology. After graduating, she started working in a lab studying anti-NMDA receptor encephalitis with Dr. Steven Mennerick. With wonderful friends and mentors to guide her, she discovered her love for synaptic physiology. She moved out to Oregon in 2018 to continue learning about her passions-- caring for others, and understanding a small piece of how the brain works. She is currently studying the role of endogenous opioids in modulating hippocampal circuitry. When not poking granule cells, Natasha can be found in the forest or on a mountain, flanked by her two sweet dogs, Gulab and Jamun.
-

AJ is a rising second year graduate student in the NGP. She was born and raised in Boise, ID and attended the University of Idaho for her bachelors degree in electrical engineering and her masters degree in biological engineering. AJ mostly focuses on electrophysiology in the lab but outside of the lab she likes to run, hike, bike, rock climb, surf, and get outside in any aspect.
-

Emma received her B.S. in Behavioral Neuroscience from Western Washington University, where she worked as an undergraduate researcher in the lab of Dr. Jeff Carroll to create CRISPR-mediated knockout cell lines to be used in the study of Huntington's disease. Emma's primary focus in the Schnell Lab is investigating the role of the α2δ-2 protein in neuronal circuits in the hippocampus and cerebellum. Her favorite cell type is the Purkinje cell. Outside the lab, Emma enjoys all things outdoors (running, biking, skiing...), crafting, and baking delicious treats.
-

Connie (she/her) recently graduated from Portland State University with a Bachelor’s of Science degree majoring in Biology and minoring in Psychology and Neuroscience, as well as on the Pre-Medicine track. She first found her passion for research as a Freshman at PSU through the BUILD EXITO program where she had the opportunity to do trauma research with Dr. Martin A. Schreiber at OHSU. In addition, she published her University Honors Thesis in collaboration with Dr. Cyndi Williams on sleep outcomes after traumatic brain injury in children using actigraphy and questionnaires. Fascinated with research and wanting to be more hands on, she joined Eric Schnell’s lab in 2024 as a Research Assistant 2 studying the timecourse of enkephalin expression in the dentate gyrus after seizures. While she is not in the lab, she enjoys traveling, exploring new food, drinking matcha, hitting the gym, and attending concerts!
-
- Ashlynn Gallagher, Research Associate, 2023-2025
- Jennifer Jahncke, Graduate Student (co-mentored with Dr. Kevin Wright, 2019-2024)
- Lucas Kuhnau, Research Associate, 2024
- Alyssa Danis, Research Associate, 2021-2024
- Ashley Anderson, Research Associate, 2022-2024
- Cory Butler, Post-doctoral Fellow, 2018-2023
- Daniel Kim, Research Associate, 2021
- Kathleen Beeson, Graduate Student, 2015-2021
- Minghui Chen, Post-Doctoral, 2020-2021
- Arielle Isakharov, Research Associate, 2018-2020
- Alexandria Wilson, Research Associate, 2018-2019
- William Hendricks, Graduate Student, 2015-2019
- Austin Peters, Resident Physician (Anesthesiology), 2016-2018
- Kunal Gupta, M.D., Ph.D., Resident Physician (Neurosurgery), 2015-2018
- Alex Lasseter, Rotation Student
- Laura Villasana, Ph.D., Post-Doc, 2012-2015
- Kayla Lanker, Summer Research Intern, 2018
- Ada Zhang, Summer Research Intern, 2017
- Yang Chen, Summer Research Intern, 2016
- Raluca McCallum, Summer Research Intern, 2013
- Kristine Kim, Summer Research Intern, 2012
- Thomas Long, Summer Research Intern, 2011
- Neelay Pandit, Summer Research Intern, 2010
- Emma Jeffries, Summer Research Intern, 2009
Publications
-
Selected Publications
- Jahncke, J.N., Schnell, E., and Wright, K.M. (2025) Distinct functional domains of Dystroglycan regulate inhibitory synapse formation and maintenance in cerebellar Purkinje cells. Communications Biology, 8(1):878.
- Danis, A. B., Gallagher, A. A., Anderson, A. N., Isakharov, A., Beeson, K. A., and Schnell, E. (2024) Altered hippocampal activation in seizure-prone CACNA2D2 knockout mice. eNeuro, 11(5):0486-23.2024.
- Jahncke, J.N., Miller, D.S., Krush, M., Schnell, E., and Wright, K.M. (2024) Inhibitory CCK+ basket synapse defects in mouse models of dystroglycanopathy. eLife, 87965.3.
- Butler, C. R., Westbrook, G. L., and Schnell, E. (2022) Adaptive mossy cell circuit plasticity after status epilepticus. Journal of Neuroscience, 42(14): 3025-3036.
- Beeson, K. A., Westbrook, G. L., and Schnell, E. (2022) α2δ-2 is Required for Depolarization-induced Suppression of Excitation in Purkinje cells. Journal of Physiology, 600(1): 111-122.
- Schnell, E., Karamooz, E., Harriff, M.J., Yates, J.E., Pfeiffer, C.D., and Smith, S.M. (2021) Construction and Validation of an Ultraviolet Germicidal Irradiation System using Locally Available Components. PLOS ONE, 16(7): e0255123.
- Beeson, K. A., Beeson, R., Westbrook, G. L., and Schnell, E. (2020) α2δ-2 protein controls structure and function at the cerebellar climbing fiber synapse. Journal of Neuroscience, 40(12): 2403-2415.
- Gupta, K. and Schnell, E. (2019) Neuronal network remodeling and Wnt pathway dysregulation in the intra-hippocampal kainate mouse model of temporal lobe epilepsy. PLOS One, 14(10): e0215789.
- Hendricks, W. D., Westbrook, G. L., and Schnell, E. (2019) Early detonation by sprouted mossy fibers enables aberrant dentate network activity. Proceedings of the National Academy of Sciences, U.S.A., 116(22): 10994-10999.
- Villasana, L. E., Peters, A. J., McCallum, R., Liu, C., and Schnell, E. (2019) Diazepam inhibits post-traumatic neurogenesis and blocks aberrant dendritic development. Journal of Neurotrauma, 36: 2454-2467.
- Chatzi, C., Zhang, Y., Hendricks, W. D., Chen, Y., Schnell, E., Goodman, R. H. and Westbrook, G. L. (2019) Exercise-induced enhancement of synaptic function triggered by the inverse BAR protein, Mtss1L. eLife 8: e45920.
- Peters, A. J., Villasana, L. E., and Schnell, E. (2018) Ketamine alters hippocampal cell proliferation and improves learning in mice after traumatic brain injury. Anesthesiology, 129, 278-295.
- Hendricks, W. D., Chen, Y., Bensen, A.L., Westbrook, G. L., and Schnell, E. (2017) Short-term Depression of Sprouted Mossy Fiber Synapses from Adult-born Granule Cells. Journal of Neuroscience, 37(23):5722-5735 and Cover Illustration.
- Villasana, L. E., Kim, K. N., Westbrook, G. L., and Schnell, E. (2015) Functional integration of adult-born hippocampal neurons after traumatic brain injury. eNeuro, 2(5) e0056-15.
- Villasana, L. E., Westbrook, G. L., and Schnell, E. (2014) Neurologic impairment following closed head injury predicts post-traumatic neurogenesis. Experimental Neurology, 261: 156-162.
- Schnell, E., Long, T. H., Bensen, A. L., Washburn, E. K., and Westbrook, G. L. (2014) Neuroligin-1 knockdown reduces survival of adult-generated newborn hippocampal neurons. Frontiers in Neuroscience, 8(71): 1-7.
- Schnell, E., Bensen, A. L., Washburn, E. K., and Westbrook, G. L. (2012) Neuroligin-1 overexpression in newborn granule cells in vivo. PLoS One, 7(10): e48045.
- Shipman, S. L., Schnell, E., Chen, B. S., Hirai, T., Roche, K. W. and Nicoll, R. A. (2011) Functional dependence of neuroligin on a new non-PDZ intracellular domain. Nature Neuroscience, 14(6): 718-26.
- Luikart, B. W., Schnell, E., Washburn, E., Bensen, A. L., Tovar, K. R. and Westbrook, G. L. (2011) Pten knockdown in vivo increases excitatory drive onto dentate granule cells. Journal of Neuroscience, 31(11): 4345-54.
- Schnell, E., Sizemore, M., Karimzadegan, S., Chen, L., Bredt, D. S., and Nicoll, R. A. (2002) Direct interactions between PSD-95 and stargazin control synaptic AMPA receptor number. Proceedings of the National Academy of Sciences, U.S.A., 99: 13902-13907.
- El-Husseini, A. E., Schnell, E., Dakoji , S., Sweeney, N., Zhou, Q., Prange, O., Gauthier-Campbell, C., Aguilera-Moreno, A., Nicoll, R. A., and Bredt, D. S. (2002) Synaptic plasticity regulated by palmitate cycling on PSD-95. Cell, 108: 849-863.
- Schnell, E. and Nicoll, R. A. (2001) Hippocampal synaptic transmission and plasticity are preserved in myosinVa mutant mice. Journal of Neurophysiology, 85(4): 1498-1501.
- El-Husseini, A. E.*, Schnell, E.*, Chetkovich, D. M., Nicoll, R. A., and Bredt, D. S. (2000) PSD-95 involvement in maturation of excitatory synapses. Science, 290(5495): 1364-1368.
Funding
- NINDS
- Department of Veteran’s Affairs
- Department of Defense
