Acute Lung Injury
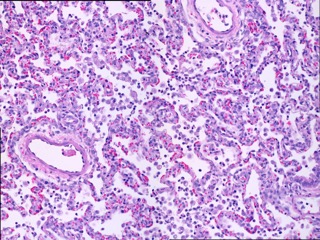
Acute lung injury is a significant contributor to morbidity and mortality in military and civilian settings. On the battlefield acute lung injury can be induced by trauma, blast injury, inhalation of noxious substances, exposure to natural or weaponized biologic agents, radioactive substances, or result as a consequence of shock from severe bleeding. In the civilian population it has been estimated that acute lung injury is associated with a 39% mortality rate with 190,600 new cases in the US yearly leading to 74,500 deaths and 3.6 million hospitalizations daily.
Systemic infection and its complications including acute lung injury and multiple organ dysfunction syndrome are important causes of morbidity and mortality in this patient population. Current effective treatment strategies for both acute lung injury and sepsis are lacking. Over the past several years our collaborators have evaluated vascular delivery of mesenchymal stromal cells (MSCs), a progenitor cell type, to the lung for the treatment of acute lung injury in rodent models, and have shown MSCs to have a pronounced therapeutic reduction in endotoxin-induced pulmonary inflammation and vascular permeability.
Currently we are developing a large animal (porcine) model of acute lung injury from inhaled endotoxin. We will use this model to perform pre-clinical studies of mesenchymal stromal cell therapy for acute lung injury that are necessary to translate this mesenchymal stromal cell therapy to military and civilian acute lung injury patients. If mesenchymal stromal cells reduce pulmonary vascular permeability and inflammation as anticipated, mesenchymal stromal cell therapy will be the first effective therapy available for acute lung injury.