Welcome to the Oregon Hearing Research Center
The Oregon Hearing Research Center (OHRC) at Oregon Health & Science University is one of the world's leading hearing-science research groups. Expertise in the OHRC spans from basic to translational science, and from the inner ear to the auditory cortex. The research focus of OHRC investigators ranges from fundamental mechanisms of hearing to development of therapies for hearing loss.

Their work has transformed the field of auditory research, and in particular have provided important advances in the study of cochlear mechanics and blood flow, how the ear's sensory cells carry out mechanotransduction, and the fine tuning of membrane properties and synapses that carry out computations in the auditory system.
In the News...
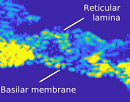
Featured Article:
Discovery of inner ear function may improve diagnosis of hearing impairment -
Science Daily
Alfred L. Nuttall, Anthony J. Ricci, George Burwood, James M. Harte, Stefan Stenfelt, Per Cayé-Thomasen, Tianying Ren, Sripriya Ramamoorthy, Yuan Zhang, Teresa Wilson, Thomas Lunner, Brian C. J. Moore, Anders Fridberger. A mechanoelectrical mechanism for detection of sound envelopes in the hearing organ. Nature Communications, 2018; 9 (1) DOI: 10.1038/s41467-018-06725-w
What's trending...
The 2024-2025 OHRC Journal Club
The OHRC Journal Club begins fall 2024 and meets most
Tuesdays, 4:00-5:30 pm in HRC 405 Seminars and Events