Biomaterials

Biomaterials for cardiovascular applications
Biomaterials for cardiovascular applications remain inadequate to meet the rigorous demands of blood contacting devices. Biomaterials must have considerable mechanical integrity to support handling, implantation, and the functional demands of the cardiovascular system, while also integrating with the biology of the surrounding area to prevent clotting and device failure. While tissue-engineered biologics or xenographs have been proposed or used, they remain an expensive and limited alternative. We aim to create devices with artificial or commercialized materials that can incorporate biologically into the native tissue.
Stents are a prominent and critical component of care for cardiovascular patients. However, the permanence of current materials, raises issues when reintervention is required. Biodegradable metals have great potential as stent materials, when mechanical and corrosive properties are optimized. Our lab is examining the biological characteristics of these biodegradable metals to understand their blood reactivity and cellular healing responses.
Thrombosis testing of biodegradable metals
Little work has examined the thrombogenicity of the metals in the absence of anticoagulants or antiplatelet therapies. While these therapies are effective in preventing biomaterial thrombosis, they present risks to patients, including an increased risk of bleeding. This work used a clinically relevant ex vivo thrombosis model to quantify thrombosis on pure metals and alloys. Our goal was to thoroughly evaluate thrombosis, including platelet attachment, fibrin accumulation, and platelet markers on metal wires exposed to whole, flowing blood.
Potential of PVA as a biocompatible material
Improvements to small diameter vascular grafts are of interest for the treatment of both coronary and peripheral artery disease. While autologous replacement is the preferred method of vascular grafting, an estimated 20% are not suitable for this method and require an artificial material. However, these synthetic grafts fail at small diameters. Current clinical vascular graft materials suffer from a compliance mismatch, which is a known cause of intimal hyperplasia and graft failure. Poly(vinyl alcohol) (PVA) is a biocompatible material, which due to the tunable material properties, can be designed to have the mechanical properties of native arteries; however, the PVA remains biologically inert, limiting the integration with the native endothelial layer. Surface modifications of PVA, including biochemical coatings and topographical cues, have the potential to improve native endothelial cell (EC) coverage, while maintaining a non-thrombogenic surface.
Pre-seeding devices with endothelial cells
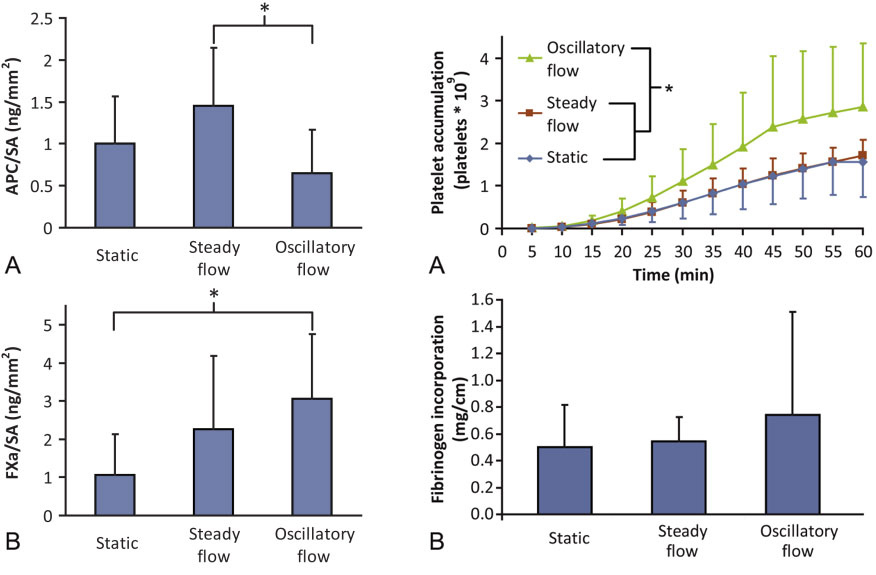
Another approach to improve current graft materials are to pre-seed devices with endothelial cells. Considerable work has been accomplished toward modifying 4 mm inner diameter ePTFE grafts with endothelial outgrowth cells. We have examined cell-seeded devices both in vitro and in vivo and observed changes to the cell phenotype with fluid pre-treatment of steady and oscillatory flow. Oscillatory flow induces significant changes in the expression of pro- and anti-thrombotic proteins (left, below) which results in a significant increase in platelet adhesion (right, below). Understanding cell phenotype, migration, and extracellular matrix production are critical components toward improving the biological integration of vascular grafts.

An overview of chronic deep venous insufficiency (CDVI)
Chronic deep venous insufficiency (CDVI) of the legs is a disease caused by absent or incompetent venous valves. CDVI occurs in up to 5% of the western population. Current treatments for CDVI are limited. Compression therapy, a palliative treatment used to control CDVI symptoms, does not address the underlying cause of the disease. Surgical repair of deep vein valves or valve transplantation is useful for only selected patients with primary CDVI, while patients with secondary CDVI are not good candidates for surgery due to residual thrombus throughout the vein.
Percutaneous or endovascular transcatheter valve implantation
A promising treatment for CDVI that is being evaluated by our research team is percutaneous or endovascular transcatheter valve implantation. This treatment involves the insertion of a bioprosthetic venous valve, designed to prevent venous reflux. Our primary biomaterial of interest for this application is small intestinal submucosa (SIS), which has been decellularized and shown to support cell growth by many researchers for different applications. Our early prototypes of these valves remained functional for 5 weeks in an animal model and in 15 patients with CDVI. However, after this time period, neointimal hyperplasia formed on the SIS leaflets, which limited their flexibility and function. Previous studies showed that leaflets lined with pre-seeded endothelial cells (ECs) prevented intimal hyperplasia from forming on the leaflets and enhanced valve functionality, suggesting the importance of ECs in preventing intimal hyperplasia. We study the effects of surface modifications on the valve leaflets to both capture circulating endothelial outgrowth cells (EOCs) and improve the function of the cells to prevent adverse remodeling responses.