Projects
Summary
Mechanisms of cell migration during embryogenesis; model systems analysis of neuronal migration in vivo; role of Amyloid Precursor Proteins as regulators of neuronal motility in the developing and aging nervous system; novel mechanisms of Ephrin-Eph receptor signaling and the control of Src-dependent neuronal guidance; Isoform-specific functions of Ig-CAM receptors in neuronal migration and axon outgrowth during embryonic development.
Focus
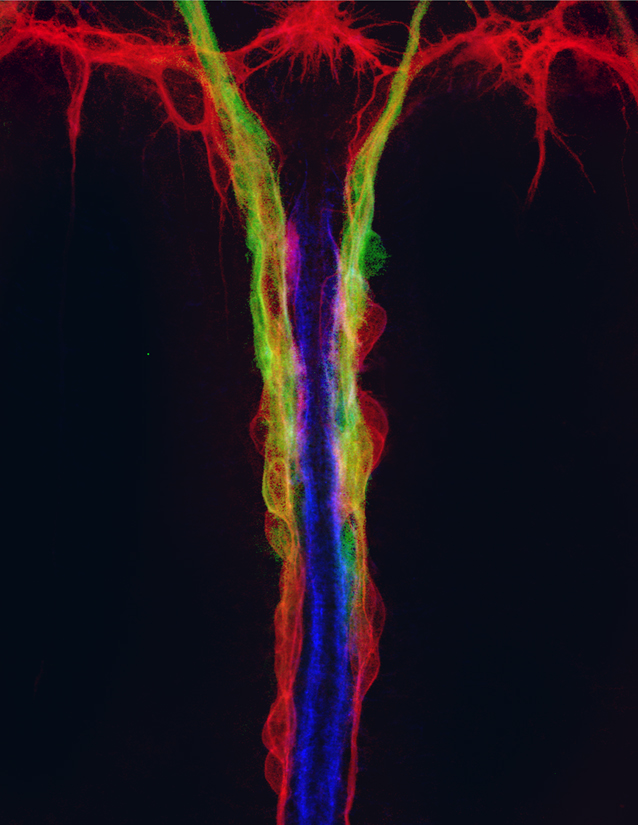
Our research is focused on the role of disease-associated genes in the control of cell migration, with an emphasis on the nervous system. During the formation of the nervous system, most neurons must migrate over extensive distances to reach their correct locations. Errors in migration can give rise to serious congenital brain defects and lead to a variety of neurological diseases, including mental retardation and epilepsy. These same transduction pathways continue to be important in the adult brain, and their disruption may play important roles in a variety of neurodegenerative conditions. What are the molecular mechanisms that regulate this process? How do neurons integrate positive and negative stimuli into coherent changes in their behavior? And how do specific disruptions in migration impact the form and function of the nervous system? Because the complexity of mammalian brain has made answering these questions difficult, we have developed a “model systems” approach, exploiting the relative simplicity of insect nervous systems to investigate the control of neuronal migration within developing embryos. This strategy allows us to explore the role of evolutionarily conserved guidance receptors and ligands that guide motile neurons in both invertebrates and vertebrates, and that may be defective in a variety of diseases affecting the human brain. Using embryonic preparations of Manduca sexta (tobacco hornworm) that permit direct access to the nervous system, we can inject individual neurons with fluorescent markers for time-lapse imaging, synthetic RNAs to induce the expression of candidate genes, and antisense constructs to inhibit gene expression at specific times during development. As a complementary strategy, we use genetic manipulations in Drosophila to test the role of specific guidance receptors and signal transduction pathways regulate neuronal guidance and differentiation in developing embryos. In addition, we employ complementary in vitro assays (including primary neuronal cultures and neuroblastoma cells) to delineate specific transduction pathways that control cell motility and guidance.
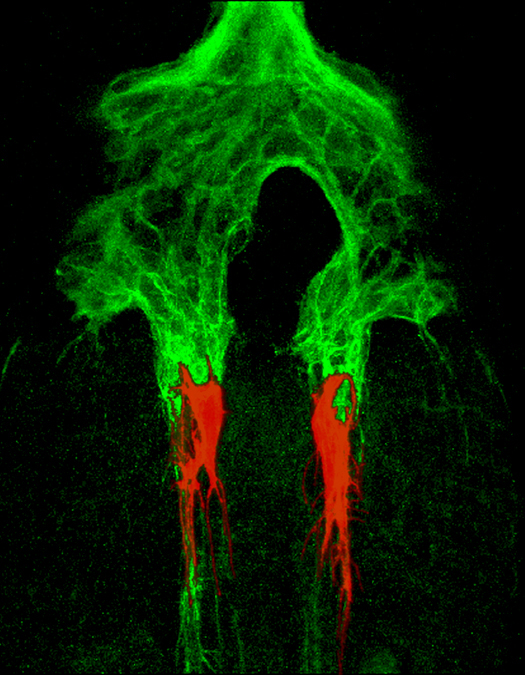
A current emphasis is the role of Amyloid Precursor Proteins (APPs) in regulating neuronal motility, a function that may be perturbed in Alzheimer’s disease. In collaboration with faculty in the departments of Neurology, Pathology, and Genetics, we have established an integrated suite of bioassays to test compounds that may ameliorate amyloid toxicity. A second project addresses novel signaling mechanisms of GPI-linked Ephrins in response to Eph receptors (originally identified as oncogenes), and their modulation of Src family kinases during development and tumor invasiveness. Yet another class of guidance receptors called Ig-CAMs promotes neuronal development in the both the insect and vertebrate nervous system, and we have discovered that different versions of the same Ig-CAM (Fasciclin II) regulate distinct aspects of migration and axon outgrowth by interacting with previously unrecognized signaling pathways. By using a combination of model systems with different experimental advantages, we can explore the role of evolutionarily conserved mechanisms that control neuronal migration, providing the framework for testing how similar signaling mechanisms may be disrupted in developmental disorders that affect the mammalian nervous system.