DDSI Margin Assessment
Intraoperative Margin Assessment using Dual Probe Difference Specimen Imaging
Intraoperative Cancer Margin Assessment
The holy grail of cancer surgery is the removal of the entire tumor with tumor margins that are negative for cancer cells, which is positively correlated with cancer cure. While negative margins are always the goal of curative cancer surgery, intraoperative margin assessment continues to challenge surgeons as their conventional tools for margin assessment include white light visualization and palpation. Lumpectomy is conventional surgical therapy for in situ breast cancer, where the resected tumor is sent to pathology for margin assessment, which is completed a few days after the surgical procedure. Unfortunately, current intraoperative margin assessment tools for breast cancer are inadequate and positive margin status is found in about 50% of cases, requiring additional surgery.
How can fluorescence image-guided surgery improve intraoperative margin assessment?
Systemic administration of fluorescent contrast agents is attractive, since it enables imaging guidance within the surgical cavity, however the availability of clinically approved contrast agents is limited and securing approval for new agents with appropriate safety profiles is a long and challenging process. Topical application of tumor-specific fluorescent probes is a conceptually-simple approach to identify tumor in specimen margins and an appealing alternative to the difficulties of in vivo contrast agent administration. However, early efforts to deploy this simple strategy produced severe non-specific uptake of targeted fluorescent probes in normal tissue, particularly in surrounding adipose tissue, a main component of resected breast specimens. Thus, simple, single-agent staining of excised tissue specimens, including tissues from lumpectomy resulted in poor diagnostic performance.
Dual Probe Difference Specimen Imaging for Intraoperative Breast Cancer Margin Assessment
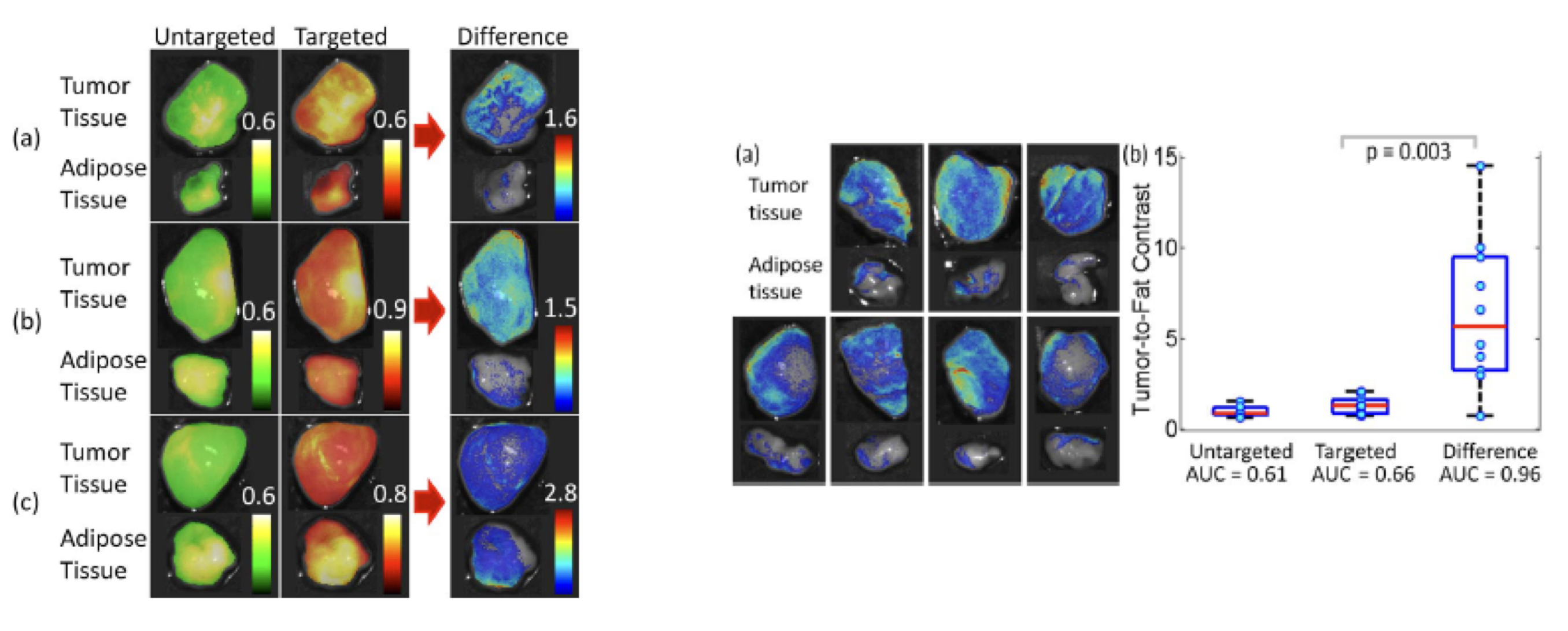
To address this, we have shown that non-specific uptake can be quantified and removed by including a second non-specific companion imaging probe in the staining solution and the targeted and non-targeted images together to improve cancer detection. In this paradigm, each probe is labeled with a spectrally-distinct fluorophore, enabling the specific and non-specific probe distribution to be assessed with multi-color imaging. The targeted and untargeted probe pairs are chosen to have similar molecular weights in an attempt to ensure similar tissue transport kinetics of both agents. Therefore, the normalized difference between the targeted and untargeted probes' fluorescence emphasizes the difference between each probe's uptake, enhancing the signal from the tumor biomarker-targeted probe. The resulting image is thus a direct representation of the targeted probe's specific binding to the tumor biomarker. We have termed this approach Dual-Probe Difference Specimen Imaging (DDSI) and are currently optimizing staining methodologies and probes for clinical translation of this technique in collaboration with Dr. Scott Davis at Dartmouth College.
Intraoperative Margin Assessment Figure
Acknowledgments
This research is funded by the Collins Medical Trust, the V Foundation for Cancer Research and the National Cancer Institute.