Pronuclear Microinjection Background
Background
The first transgenic animals were produced more than 30 years ago (Jaenisch &Mintz, 1974;Gordon et al., 1980) and now transgenic technology is successfully and broadly used in different fields of biomedical research. Animals can be defined as transgenic when they have integrated foreign DNA into their germ line as a consequence of experimental introduction of DNA (Palmiter &Brinster,1985). Although several approaches exist to introduce foreign DNA into the mammalian genome, the majority of transgenic mice are produced by microinjecton of plasmid DNA into the pronucleui of fertilized eggs. Traditionally to achieve stable integration and expression a transgene is injected as a several kilobase fragment of plasmid DNA. Volume of injected DNA is approximately 1-2 picoliters (pl) at a concentration of 1-2 ng /ml. As a result, it can integrate into random regions of different chromosomes as a concatemer (several copies of transgene) and thus the founders may have different levels of transgene expression. About 10-15% of the mice we have produced from DNA microinjection have tested positive for the transgene. The initial mice that carry the transgene are called 'founder' (or Fo) transgenic animals. The F1 offspring result after mating Fo and wildtype mice and the transgene containing individuals will give origin for the new transgenic line.
Many different modifications of transgenic technology have been developed. For example, to achieve single copy integration of a transgene and expression at physiological level in the appropriate cell types a transgene together with regulatory elements can be delivered to the mouse genome as a Bacterial Artificial Chromosome (BAC) or Yeast Artificial Chromosome (YAC) containing several hundred kilobases from mouse or human genome (Giraldo, Montoliu, 2001;Van Keuren et al., 2009). Since the majority of injected genes are expressed, this technique is generating exciting opportunities to study differential gene function, abnormal gene regulation in malignant disease and mutations of genes cause developmental effect. Moreover, the use of for example, tet-inducible (Freundlieb et al., 1997;Fedorov et al., 2000) and/or Cre/LoxP conditional (Schönig et al., 2002;Wang, 2009) transgenic systems provides an ability to controlling mammalian gene expression in a temporal and spatial manner particularly during development and oncogenesis (for more info, see Useful Links and References).
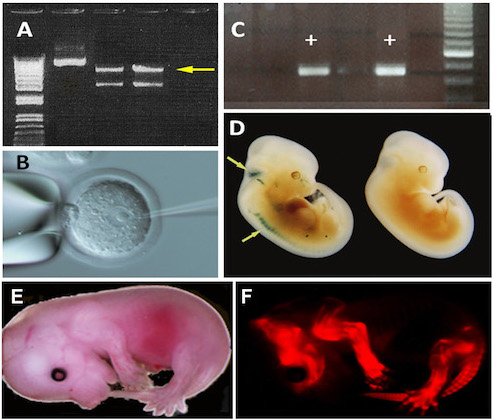
Generation of transgenic mice. (A) Isolation of the 7 kb transgene (yellow arrow) on an agarose gel after digestion of a plasmid by HindIIIand XhoI enzymes. (B) MicroinInjection of a transgene into the pronucleus of a fertilized mouse egg. (C) PCR identification of transgenic founders (+) among newly generated progeny. (D) Tet-inducible expression of a beta-gal reporter gene in the roof of the hindbrain and spinal ganglia (yellow arrows) of tTACMV/NZL double transgenic embryos at E12.5. (E) Lateral overview of a "Cherry" transgenic embryo at E15.5. (F) "Cherry" expression in the skeleton of the embryo. The transgenic mice were produced for Dr. R. Schweitzer (Shriners Hospital, OHSU, Portland, OR).