Powers Lab Current Projects
Death Receptor Project:
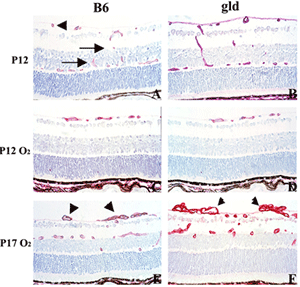
Activation of specific death receptors by FasL or Trail leads to conformational changes in the intracellular domain of the receptors that cause recruitment of the adaptor protein FADD and caspase-8 leading to activation of caspase-8 proteolytic activity and apoptosis. We are investigating the role of endothelial cell apoptosis in vascular tuft regression through the use of FasL mutant mice (gld), TRAIL KO mice, and injection of soluble FasL. As seen in the figure, the absence of function FasL does not alter vaso-obliteration (D) as compared to control B6 mice (C) after 5 days of hyperoxia exposure (P12). In contrast, when the mice are recovered in room air, the gld mice exhibit increased retinal neovascularization on P17 (F) compared to controls (E). The gld mice also had reduced apoptosis in the vascular tufts (not shown), suggesting that FasL/Fas interactions (apoptosis) are important in controlling retinal neovascularization.
Role of Microglia/Macrophages in Vascular Regression:
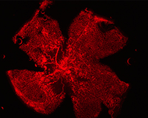
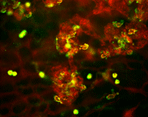
We are using genetically altered mice to test the hypothesis that macrophages and microglia play a significant role in the regression of retinal neovascularization (NV) by promoting apoptosis. The figure on the left exhibits retinal NV (lectin stained, red) from a P17 retina after hyperoxia- induced retinopathy. The figure on the right demonstrates the co-localization of macrophages/microlia (green) with preretinal tufts (red). Through the use of MCP-1 KO mice and clodronate depletion, we will determine if reduced numbers of macrophages/microglial cells results in altered endothelial apoptosis.
EphrinB2 and EphB4 Receptor Project:
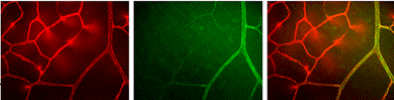
EphrinB2 and EphB4 receptors have emerged as key regulators of vascular development and are primarily expressed by endothelial cells of arteries and veins, respectively. These molecules are also associated with postnatal angiogenesis. We have used soluble forms of these molecules to reduce retinal NV in the mouse model of oxygen-induced retinopathy. We are now actively examining the expression of these molecules in retinal vascular development and pathologic angiogenesis through the use of ephrinB2- and EphB4-lacz+/- mice. The figure below shows lectin stained retinal vessels (red, left), ephrinB2 labeled retinal arterioles (green, middle), and minimal capillary overlap (right) in the normal mouse retina on postnatal day 12.
RTEF-1 Project:
Our lab is providing the expertise of the mouse model of OIR to the Stout Lab at OHSU. The Stout lab is investigating the transcriptional enhancer factor 1-related (RTEF-1) and its isoforms in the regulation of VEGF expression under hypoxic conditions in vitro (endothelial cell culture) and in vivo (mouse model of OIR).
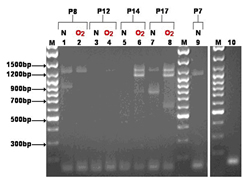
Novel isoforms for RTEF-1 exit within mouse neural retina and differ between control and an oxygen induced retinopathy model (OIR) depending on the day of postnatal (P) development (from P7-P17).
More information
Contact us
Departments of Pediatrics and Ophthalmology
Mail Code: L467
3181 S.W. Sam Jackson Park Road
Portland, OR, 97239
Lab Phone: 503-494-3371
Pulmonary Office: 503-494-8023
Lab Fax: 503-494-6875
Email: Mike Powers & Mike Davies