Duvoisin Lab
The objectives of the laboratory are to study the structure, distribution, and function of G protein-coupled metabotropic glutamate receptors (mGluRs). Because mGluRs modulate, as opposed to mediate, synaptic transmission, compounds acting on mGluRs are being investigated as therapies and are in clinical trials for neurological and psychiatric diseases such as central pain, drug addiction, excitotoxic neuronal injury following stroke, Parkinson's disease, Alzheimer's disease, anxiety, and schizophrenia.
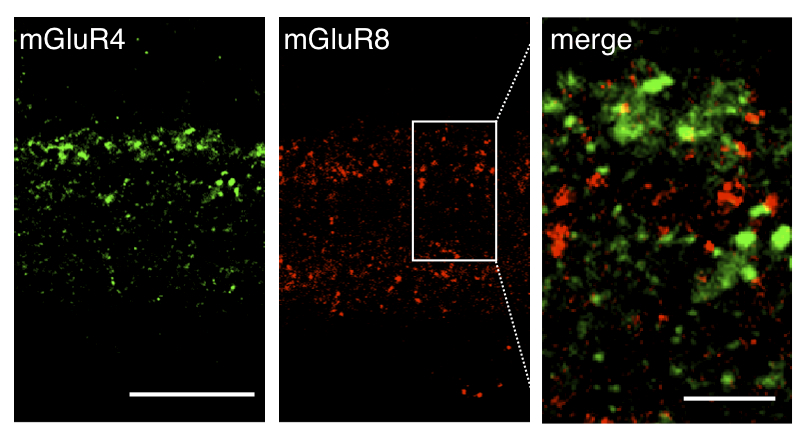
The cloning of neurotransmitter receptors led to the unexpected finding that they occur as numerous subtypes encoded by gene families. The question of how this newfound molecular diversity relates to the complexity of neuronal cell types and circuitry was clearly interesting and important, both as a means to understand how neural circuits process information, and because drugs specific for defined receptor subtypes and neural circuits could potentially be used as therapeutics. Because the development and the circuitry of the retina have been uniquely well studied, we believe the retina is an ideal model to examine the relationship between mGluR diversity and neural cell types and circuits (Fig. 1). For publications on this project see J Comp Neurol.
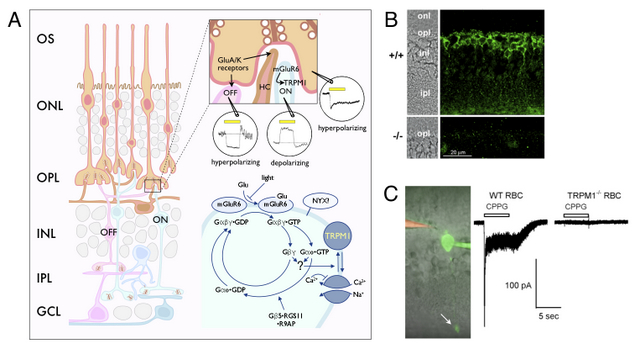
In the retina, photoreceptors transduce light stimuli into a regulated synaptic release of glutamate. There are two types of second-order neurons, the ON- and OFF-bipolar cells, which respond in opposite way to the light-regulated synaptic glutamate (Fig. 2A). OFF-bipolar cells express AMPA/kainate ionotropic glutamate receptors. In contrast, ON-bipolar cells express mGluR6. Recently, we have initiated proteomic and genetic approaches to identify components of the signaling pathway activated by mGluR6 in ON-bipolar cells. While phototransduction has been at the forefront of research on G protein signaling and its regulation, much less is known about the mGluR6- activated signaling pathway. In collaboration with Drs. Catherine Morgans, Lane Brown (Washington State University), and Brett Jeffrey (National Eye Institute) we have found that TRPM1 is expressed in ON-bipolar cells (Fig. 2B) and required for the depolarizing response of ON-bipolar cells to light (Fig. 2C). Ongoing research aims to reconstitute the signaling pathway from mGluR6 to TRPM1. For publications on this project see PNAS and BioEssays.
Recently, we and other have found that serum from cutaneous malignant melanoma patients with melanoma-associated retinopathy contains autoantibodies against TRPM1. Our research is aimed at a better understanding of the mechanism by which these autoantibodies arise and whether the serum of CMM patients without visual symptoms may also contain autoantibodies. For recent publications on this project see PLoS ONE and Ophthalmology.
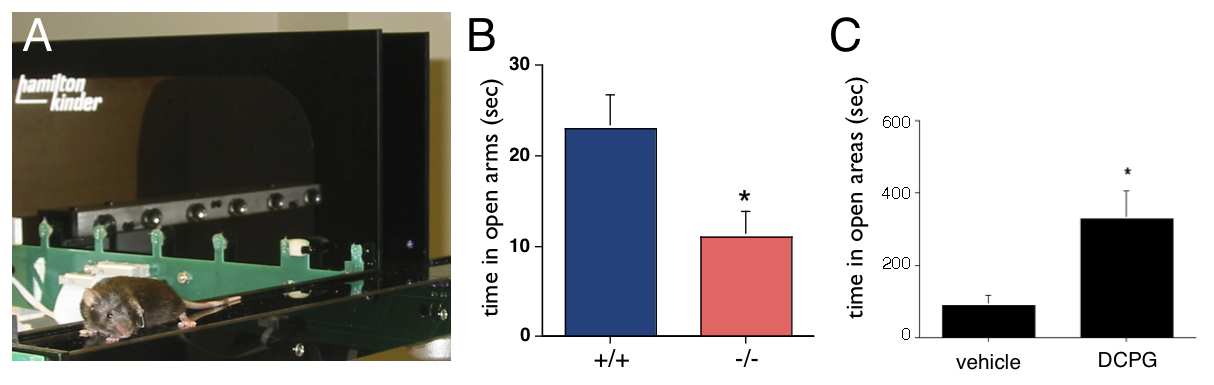
In collaboration with Dr. Jacob Raber (Behavioral Neuroscience), we have shown that mice deficient for mGluR8 have increased behavioral measures of anxiety (Fig. 3B). Further, we have found that acute administration of DCPG, a specific mGluR8 agonist, reduces anxiety-like behaviors in mice with heightened anxiety levels (Fig. 3C). We are currently also examining the roles of a related receptor, mGluR4, in anxiety-like behaviors. Our identification of mGluR4 and mGluR8 as therapeutic targets for anxiety disorders seems particularly promising for the significant portion of patients with anxiety disorders showing benzodiazepine insensitivity. For recent publications on this project see Neuropharm.